2021 in 2022 Accepted Abstracts
* Indicates selected for podium presentation
Trends and Outcomes of Salvage Lobectomy for Early-Stage Non-Small Cell Lung Cancer*
Sam Afshari, Nataniel H. Lester-Coll, MD
University of VermontBackground: There is little data describing outcomes for patients who develop local recurrences following stereotactic body radiation therapy (SBRT), a standard of care treatment option for patients with early-stage non-small cell lung cancer (NSCLC). One emerging option is salvage lobectomy. We investigated trends in the use of salvage lobectomy following SBRT sand described patient outcomes using a nationally representative sample.
Methods: This is a retrospective study using the National Cancer Database of patients with NSCLC diagnosed from 2004 to 2017. We used descriptive statistics to describe patients who underwent salvage lobectomy. Kaplan-Meier analysis was used to describe overall survival (OS) and time to lobectomy. Cox proportional modeling was used to identify factors associated with survival.
Results: We identified 295 patients who underwent salvage lobectomy. Ninety-day mortality was 0%. Median time to lobectomy was 85 months (95% CI 77 – 97). Median survival for the cohort was 49 months (95% CI 44 - 58). Squamous cell histology was associated with improved OS (HR = 1.61, P = 0.003). Factors associated with decreased OS included female sex (0.64, P = 0.007) and increased time to lobectomy (0.71, P = 0.014). OS was worse when salvage lobectomy was done after 6 months of SBRT (0.47, P = 0.007), compared to between 1 and 6 months (0.59, P = 0.049).
Conclusions: Salvage lobectomy following local failures of SBRT was associated with no perioperative mortality and favorable long-term outcomes. Our data suggests earlier intervention for treatment failures results in better OS.
Vitamin B12 Deficiency Mimicking Thrombotic Microangiopathy
David Alfonso Alejos Castillo, Rohit Singh MD, Mansour Gergi MD
University of Vermont Medical Center/Department of Hematology and Oncology
Background: Thrombotic microangiopathies (TMA) presents with microangiopathic hemolytic anemia (MAHA), thrombocytopenia. However, these features can occur in the context of vitamin B12 deficiency. Recognizing this entity is essential when evaluating these patients
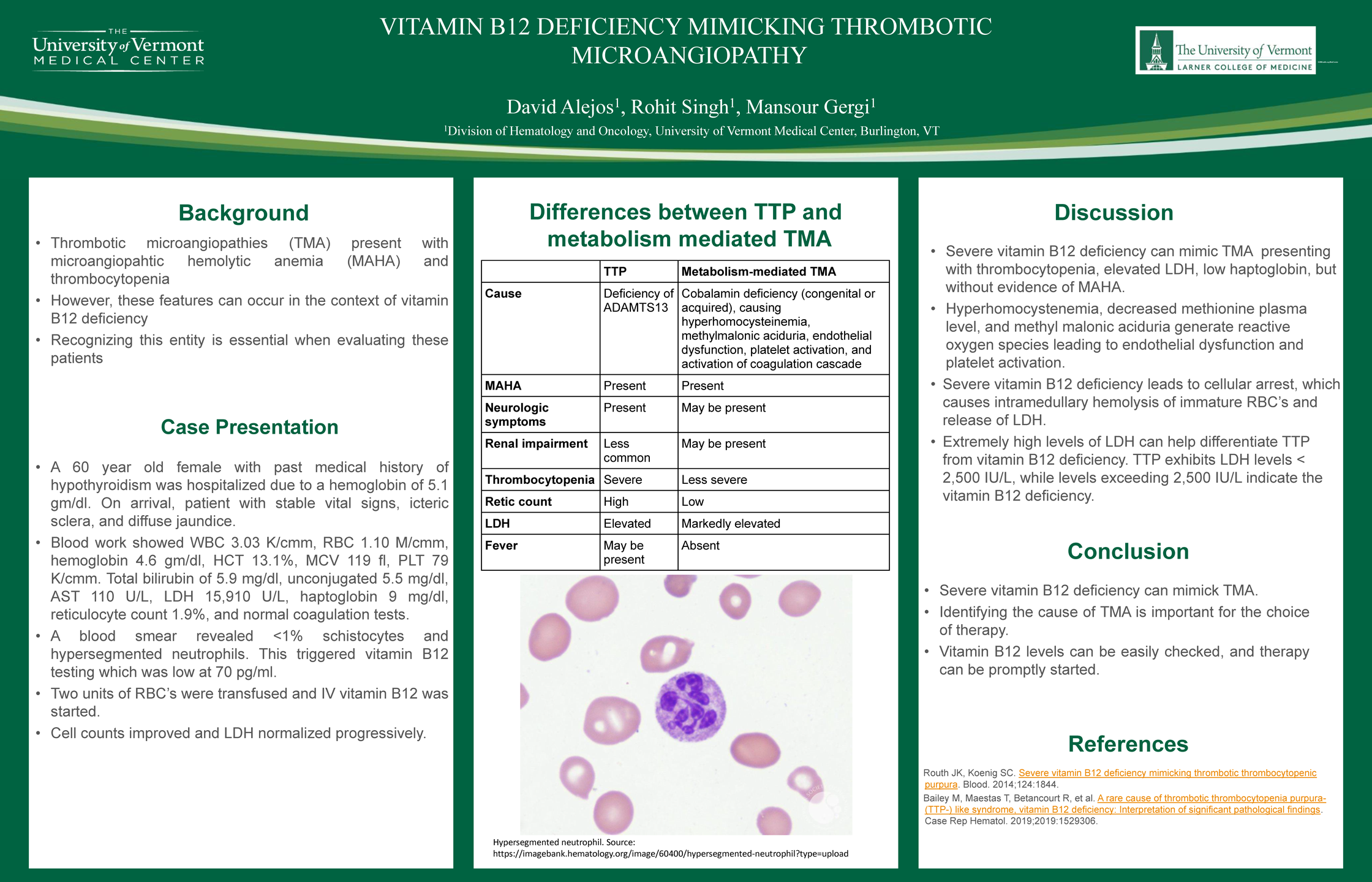
Methods: A 60-year-old female with past medical history of hypothyroidism was hospitalized due to a hemoglobin of 5.1 gm/dl. On arrival, vital signs were stable, physical exam was remarkable for icteric sclera and jaundice. A complete blood count showed WBC 3.03 K/cmm, RBC 1.10 M/cmm, hemoglobin 4.6 gm/dl, HCT 13.1%, MCV 119 fl, PLT 79 K/cmm. A total bilirubin of 5.9 mg/dl (indirect 5.5 mg/dl), AST 110 U/L, LDH of 15,910 U/L, haptoblgobin 9 mg/dl, reticulocyte count 1.9%, and normal coagulation tests. A blood smear revealed <1% schistocytes and hyper segmented neutrophils, this triggered vitamin B12 testing which was low at 70 pg/ml. Two units of RBCs were transfused and IV vitamin B12 therapy was started. Cell counts improved and her LDH normalized progressively.
Results: Severe vitamin B12 deficiency can mimic TMA presenting with thrombocytopenia, elevated LDH, low haptoglobin, but without evidence of MAHA. Hyperhomocystinemia, decreased methionine plasma level, and methyl malonic aciduria generate reactive oxygen species leading to endothelial dysfunction and platelet activation. High levels of LDH, the absence of increased reticulocyte count, and macrocytosis are highly suggestive of severe vitamin B12 deficiency.
Conclusions: Severe vitamin B12 deficiency can present as pseudo-TMA. Identifying the cause of TMA is important for the choice of therapy. Vitamin B12 levels can be easily checked, and therapy can be promptly started.
Exploring the presence of Bovine Leukemia Virus among breast cancer tumors in a rural state: a preliminary report
Stas Amato, MD, PGY-Research
University of Vermont Medical Center
Background: The bovine leukemia virus (BLV) is a causative agent for malignant lymphoma and lymphosarcomas in cattle globally. It is a deltaretrovirus with a trans-activating region of the X-gene (Tax), which causes malignancy in host cells by inhibiting DNA repair and disrupting mechanisms for the control of cellular growth. The virus is ubiquitous among U.S. dairy herds, an association between BLV DNA and human breast cancer has been demonstrated. We aim to describe investigate the presence of BLV DNA among breast cancer samples in a rural state.
Methods: We collected 31 samples of frozen breast cancer tissue that was banked between 2016-2019. Genomic DNA was purified from fresh-frozen sections, and with nested PCR assays, BLV Tax sequence primers were used to identify the presence of BLV DNA in tissue samples and known positive control samples, including locally infected bovine blood.
Results: Of the 31 breast tumor samples, the majority of tissue diagnoses were poorly differentiated invasive ductal adenocarcinoma. On PCR assay, none of the 31 tumor samples tested positive for BLV DNA.
Conclusions: The identification of a preventable environmental cause of breast cancer could have important public health indications for disease control and prevention. We did not find BLV DNA in breast cancer tumors of patients presenting to a hospital in Vermont. While our findings suggest that BLV DNA is not present in breast cancer tumors among our patient population, a larger sample size and immunohistochemistry studies could be utilized to confirm findings.
A right to left intrapulmonary shunt due to hepatopulmonary syndrome due to metastatic liver disease in GE junction adenocarcinoma.
Rahul Anil, Steve Ades
University of Vermont Medical Center
Background: Hepatopulmonary syndrome is a triad of arterial blood deoxygenation, intrapulmonary vasodilation, and liver disease. Cirrhosis and portal hypertension have been reported to be the common causes but rarely due to metastatic liver cancer from colon or breast has been reported in case reports. We report a rare presentation of hepatopulmonary syndrome in gastroesophageal adenocarcinoma metastatic to liver which has not been reported in literature yet.
Case Report: A 46-year old with a history of metastatic gastroesophageal junction adenocarcinoma on third line therapy with pembrolizumab presented with worsening shortness of breath that initially started with exertion but gradually worsened over two weeks to rest. He had no orthopnea, platypnea, or leg swelling. He was hypoxic SPO2 88% requiring 4L O2 only to saturate 90%. CT PE showed no pulmonary embolism, AVMs, consolidation, pleural effusion or pneumonitis. Abdomen/pelvis CT showed increase in size of numerous hepatic metastases, new omental nodularity with moderate ascites consistent with peritoneal carcinomatosis. Ascitic fluid SAAG >1.1, positive malignant cells suggestive of mixed picture. Synthetic function of liver was normal except chronically elevated AST/ALK. Transthoracic echocardiography with bubble study revealed bubbles in the left atrium after 6 heart beats suspicious for an intrapulmonary and not an intra-cardiac shunt. VQ scan confirmed intrapulmonary shunt. Overall, hypoxia refractory to 100% oxygen, increasing hepatic metastasis, ascites and intrapulmonary shunt was indicative of hepatopulmonary syndrome secondary to metastatic liver.
Conclusions: This emphasizes that hepatopulmonary syndrome, a less common morbidity secondary to metastatic liver lesions, should require timely identification and management due to its life-threatening outcome.
A RARE CASE OF PRIMARY APOCRINE ADENOCARCINOMA
Asad Anwar, MBBS
The University of Vermont Medical Center
Introduction: Cutaneous adenocarcinomas with apocrine features are rare and consensus guidelines do not currently exist. They typically develop in areas of high sweat gland density (1) and often mimic benign etiologies such as hidradenitis. Most present as localised disease but around 30% can be locally advanced or metastatic. (2). Prognosis in the metastatic setting is generally poor (3).
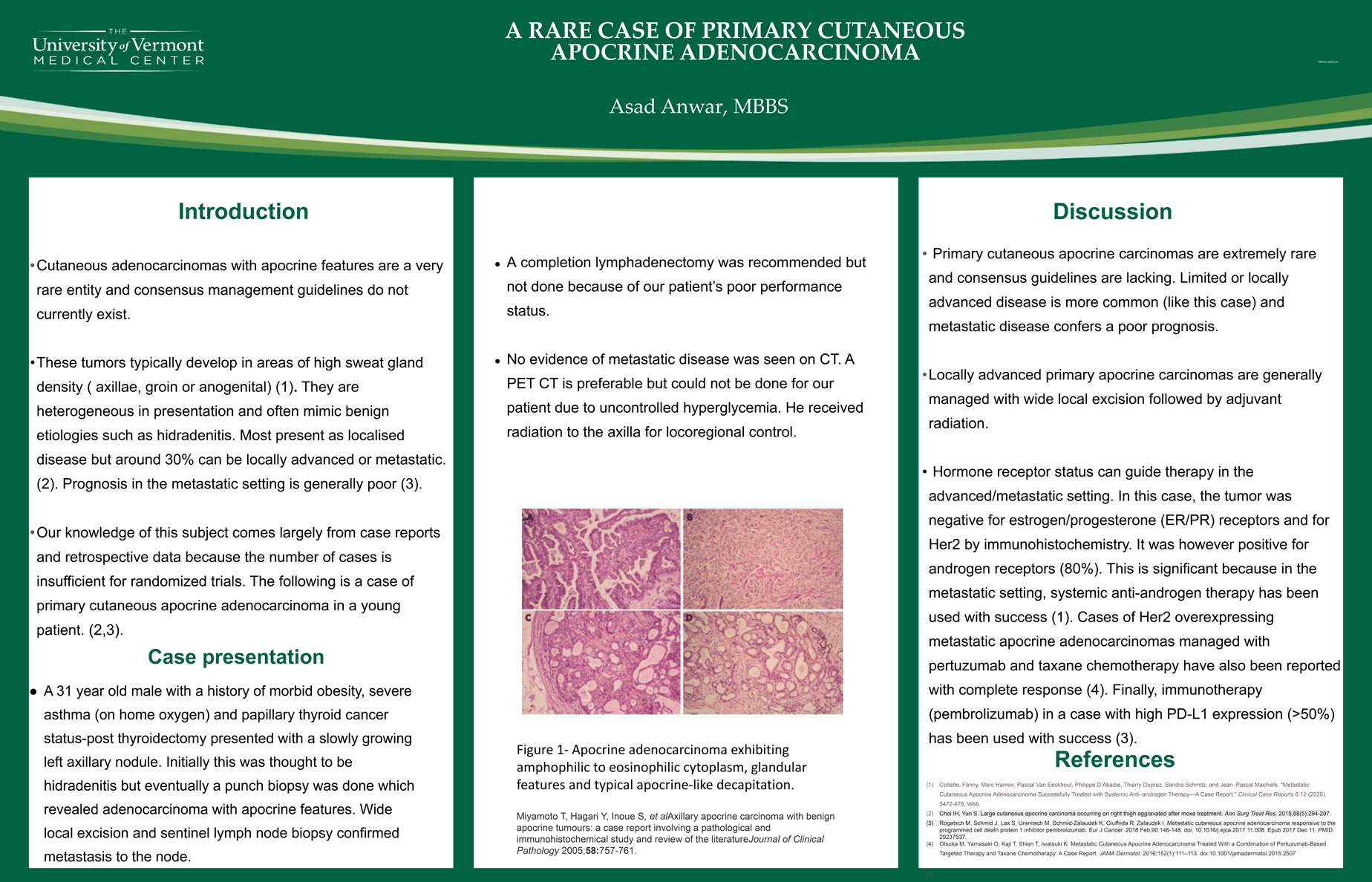
Case: A 31 year old male with a history of morbid obesity, asthma and papillary thyroid cancer presented with a slowly growing left axillary nodule. Initially this was thought to be hidradenitis but a punch biopsy revealed adenocarcinoma with apocrine features. Wide local excision and sentinel lymph node biopsy confirmed metastasis to the node. A completion lymphadenectomy was recommended but not done because of our patient’s poor performance status. No evidence of metastatic disease was seen on CT. He received radiation to the axilla for locoregional control.
Discussion: Primary cutaneous apocrine carcinomas are extremely rare and consensus guidelines are lacking. Limited/locally advanced disease is more common (like this case) and are generally managed with wide local excision followed by adjuvant radiation.
Hormone receptor status can guide therapy in the advanced/metastatic setting. In this case, the tumor was negative for estrogen/progesterone receptors and Her2 but positive for androgen receptors (80%). Systemic anti-androgen therapy has been used with success (1). Her2 overexpressing metastatic apocrine adenocarcinomas managed with pertuzumab and taxane chemotherapy have also been reported with complete response (4). Finally, immunotherapy (pembrolizumab) in a case with high PD-L1 expression (>50%) has been used with success (3).
References: (1) Collette, Fanny, Marc Hamoir, Pascal Van Eeckhout, Philippe D’Abadie, Thierry Duprez, Sandra Schmitz, and Jean‐Pascal Machiels. "Metastatic Cutaneous Apocrine Adenocarcinoma Successfully Treated with Systemic Anti‐androgen Therapy—A Case Report." Clinical Case Reports 8.12 (2020): 3472-478. Web.
(2) Choi IH, Yun S. Large cutaneous apocrine carcinoma occurring on right thigh aggravated after moxa treatment. Ann Surg Treat Res. 2015;88(5):294-297.
(3) Rogatsch M, Schmid J, Lax S, Uranitsch M, Schmid-Zalaudek K, Giuffrida R, Zalaudek I. Metastatic cutaneous apocrine adenocarcinoma responsive to the programmed cell death protein 1 inhibitor pembrolizumab. Eur J Cancer. 2018 Feb;90:146-148. doi: 10.1016/j.ejca.2017.11.008. Epub 2017 Dec 11. PMID: 29237537.
(4) Otsuka M, Yamasaki O, Kaji T, Shien T, Iwatsuki K. Metastatic Cutaneous Apocrine Adenocarcinoma Treated With a Combination of Pertuzumab-Based Targeted Therapy and Taxane Chemotherapy: A Case Report. JAMA Dermatol. 2016;152(1):111–113. doi:10.1001/jamadermatol.2015.2507
Blasting through the phases: a case of rapid progression of CML to blast phase
Odeth Barrett-Campbell, MD, Swaroopa Yerrabothala, Marc Gautier
Department of Internal Medicine, Section of Hematology & Oncology, DHMC
Background: CML is a triphasic myeloproliferative neoplasm associated with Ph chromosome and the BCR-ABL1 fusion gene, resulting in p210 BCR-ABL1 protein. The natural course of untreated CML is progression to accelerated phase and then blast phase (BP) within 2-5 years. CML-BP manifests as a lymphoid lineage in ~30% of cases and is often clinically indistinguishable from de novo Ph (+) ALL. However, the presence of p210 transcripts in B lymphoblasts, though not diagnostic, is suggestive of emergence from underlying CML.
Methods: A 71 y/o male presented with fatigue, drenching sweats, weight loss, splenomegaly, WBC 52K. Bone marrow biopsy (BMB) was consistent with CML in chronic phase with 1% blasts, p210 BCR-ABL1 transcript > 50%. He was started on imatinib 400mg daily. At 3 months, he achieved complete hematological response. However, his BCR-ABL1 remained at > 50%. Imatinib was continued and by 6 months, BCR-ABL1 improved to 20%. Three weeks later, he presented with fevers, anorexia, and WBC of 19K with 20% peripheral blasts. BMB showed Ph + B-ALL with >90% blasts; p210 BCR-ABL1 transcript >50%. TKI resistance mutation analysis by Sanger sequencing (SS), was negative. He was induced with HyperCVAD plus dasatinib. BMB after 1st cycle showed complete morphological remission; BCR-ABL positive improved to 2.12%.
Conclusions: The precise molecular mechanisms leading to the unusual rapid progression to the BP remains poorly understood. A leading theory is treatment resistance due to the acquisition of BCR-ABL1 kinase domain (KD) mutations. SS has an estimated 15 – 25% lower limit for detecting KD mutations. Correlation with clinical and other laboratory findings is crucial.
Mets to where? Axillary adenopathy at the initial presentation of advanced ovarian cancer
Raven Bennett1, Odeth Barrett-Campbell2, Gary Schwartz2
1 Geisel School of Medicine at Dartmouth, 2 Department of Internal Medicine, Section of Hematology & Oncology, Dartmouth-Hitchcock Medical Center
Background: The majority of patients with ovarian cancer present with advanced disease largely from intraperitoneal spread or local infiltration. Distant metastases via lymphatic or hematogenous infiltration is rare, which makes the presentation with breast and axillary adenopathy rather unusual.
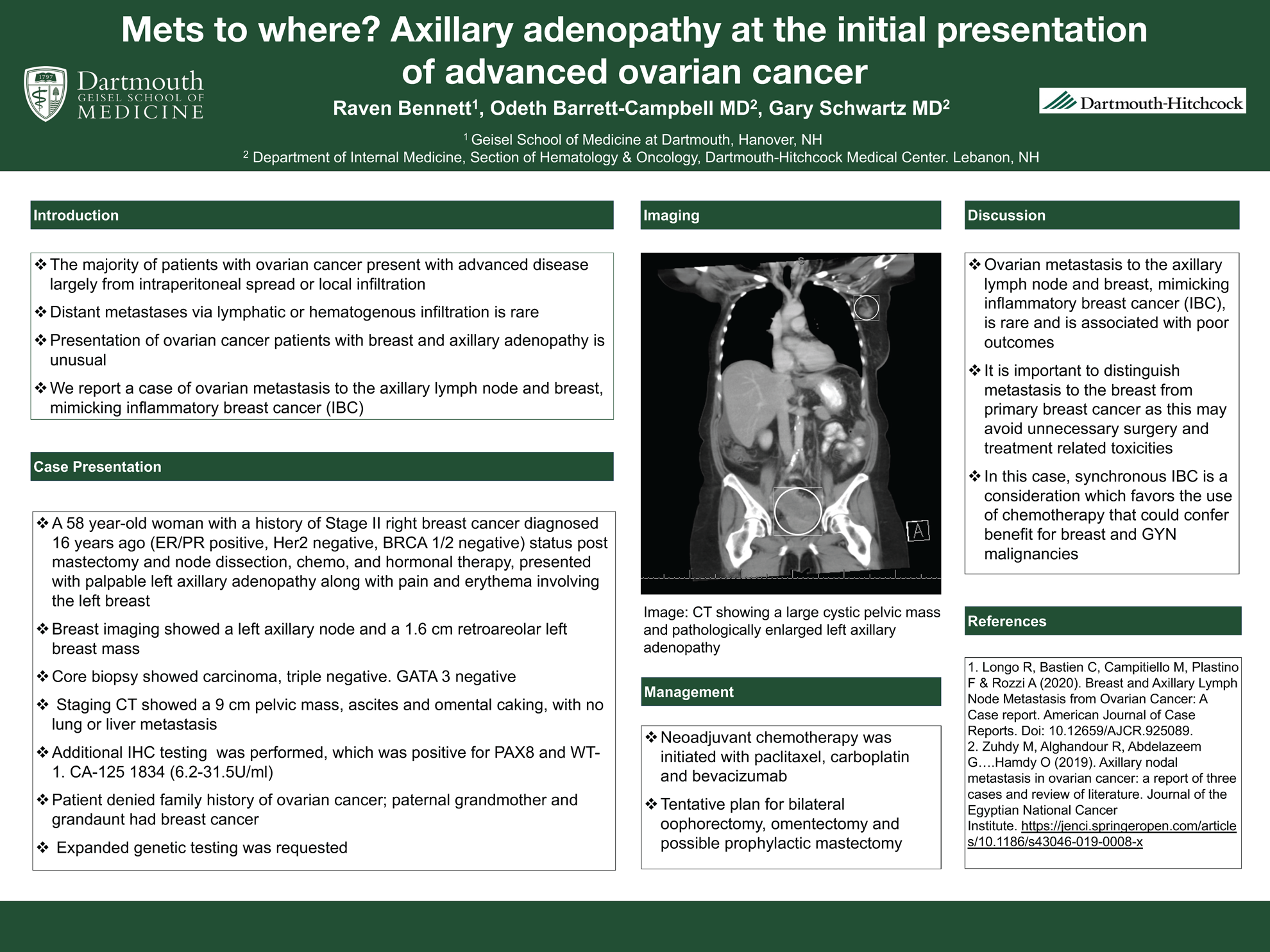
Methods: Here we present a 58 y-o woman diagnosed with Stage II right breast cancer diagnosed 16 years ago, ER/PR +ve, Her2 -ve, BRCA –ve, status post mastectomy and node dissection, chemo and hormonal therapy who presented with palpable left axillary adenopathy along with pain and erythema involving the left breast. Breast imaging showed a left axillary node and a 1.6cm retroareolar left breast mass. Core biopsy showed carcinoma, triple negative. GATA 3 negative. Staging CT showed a 9 cm pelvic mass, ascites and omental caking, no lung or liver mets, prompting additional IHC testing which was positive for PAX8 and WT-1. CA-125 1834 (6.2-31.5U/ml). She denied family history of ovarian cancer; paternal grandmother and grandaunt had breast cancer. Neoadjuvant chemotherapy with paclitaxel, carboplatin and bevacizumab followed by tentative plan for bilateral oophorectomy, omentectomy and possible prophylactic mastectomy. Expanded genetic testing requested.
Conclusions: Ovarian metastasis to the axillary LN and breast, mimicking inflammatory breast cancer (IBC), is rare and is associated with poor outcomes. It is important to distinguish metastasis to the breast from primary breast cancer as this may avoid unnecessary surgery and treatment related toxicities. In this case, synchronous IBC is a consideration which favors the use of chemotherapy that could confer benefit for breast and GYN cancers.
Understanding cancer survivorship experiences in New Hampshire (NH)
Adriane Burke, MPH, Scot Foster, BS, Whitney Hammond, MSW, MPH
NH Division of Public Health Services
Methods: A multi-pronged approach to data collection was used to understand the experiences of cancer survivors in New Hampshire.
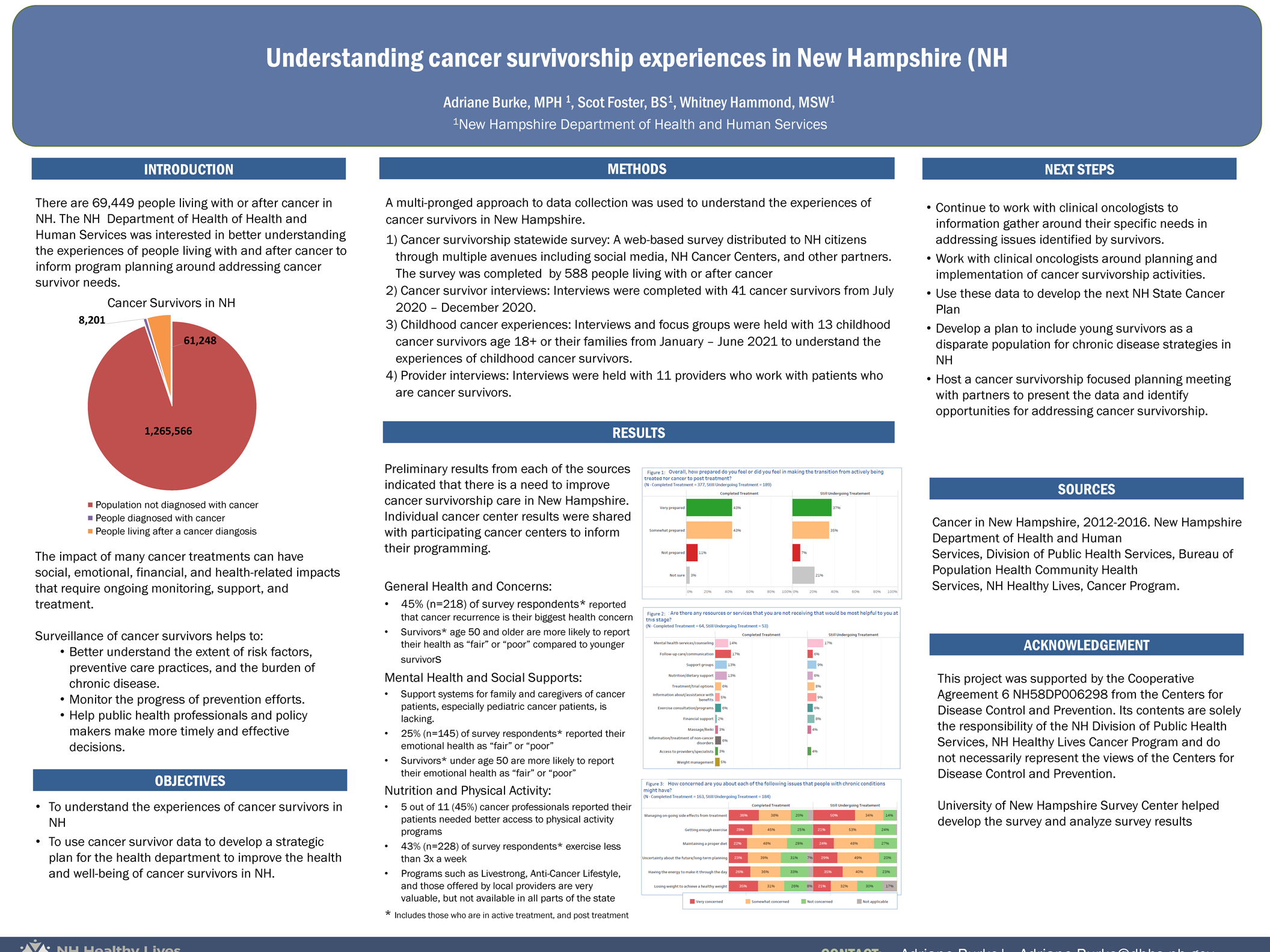
- Cancer survivorship statewide survey: A web-based survey distributed to NH citizens through multiple avenues including social media, NH Cancer Centers, and other partners. The survey was completed by 588 people living with or after cancer
- Cancer survivor interviews: Interviews were completed with 41 cancer survivors from July – December 2020.
- Childhood cancer experiences: Interviews and focus groups were held with 13 childhood cancer survivors age 18+ or their families from January – June 2021 to understand the experiences of childhood cancer survivors.
- Provider interviews: Interviews were held with 11 providers who work with patients who are cancer survivors.
Results: Results from each of the sources indicated that there is a need to improve for cancer survivorship care in New Hampshire. Individual cancer center results will be shared with participating cancer centers to inform their programming.
Conclusions: The data observed through these data collection efforts will be used to inform program planning efforts at the SDOH for cancer survivors living in New Hampshire.
Notes: This project was funded by the Centers for Disease Control and Prevention.
Enhancing CAR T-cell Therapy Care and Survivorship through the Patient Experience
Kate Caldon, RN, Lisa Wesinger RN, BMT Nurse Navigator, Judi Gentes RN, BMT Nurse Navigator, Susan Brighton NP, Dr. John Hill Jr., MD, Carissa Morton RN, Sara Smith RN, Ashley Prokopik RN
DHMC - Norris Cotton Cancer Center
Background: Chimeric Antigen Receptor (CAR) T-cell therapy is a novel cellular immunotherapy to treat advanced cancer. Dartmouth-Hitchcock Medical Center (DHMC) obtained certification in 2019 to offer this to patients in Northern New England. Amidst the ensuing COVID-19 pandemic, the first patient received this modality in April 2020. The patient/caregiver perspective obtained through recorded interviews allows first-hand insight into the lived experience, serving to illustrate the unique facets of CAR T-cell therapy.
Methods: Interpretive phenomenological analysis (IPA) was utilized for interviews with the patient/caregiver at 1 year post CAR T-cell therapy. Questions track their story from diagnosis through survivorship, creating an enhanced clinical case study. Integrating the medical/clinical background serve as a didactic platform for provider/nursing education on this complex treatment. By embedding the patient experience into the case study nursing theory is utilized to:
- Demonstrate how inviting patients to recount their story fosters emotional investment, invaluable insight and improved advocacy for the needs of the whole person.
- Illustrate the impact of the patient/caregiver relationship, ensuring autonomy and contributing to optimal outcomes and strengthen survivorship.
Results: DHMC’s first patient/caregiver team reported receiving excellent care and the opportunity to relate their lived CAR T-cell therapy “journey” was, itself, therapeutic. Qualitative themes that emerged will be discussed.
Conclusions: A positive patient experience can be used to ensure ongoing advocacy for the needs of the whole person. This format illustrates how a complex treatment can be delivered while still effectively caring for the mind, body and spirit of each unique patient/caregiver team.
Modified frailty index (mFI) in elderly patients with glioblastoma correlates with survival*
Amy Chang, Alissa A. Thomas, MD
University of Vermont Larner College of Medicine
Background: Glioblastoma carriers a poor prognosis in elderly. The mFI predicts post-surgical in-hospital mortality and shorter overall survival (OS). We applied mFI-11 to a cohort of surgically confirmed glioblastoma within our institution to examine treatment and survival patterns.
Methods: We performed a retrospective cohort study from 2016-2021 in patients 65 with glioblastoma. We applied a frailty score to each patient using the (mFI-11). We used the mFI-11 index to compare survival outcomes and NCCN recommended treatments.
Results: We identified 108 patients 65 with an EMR code for glioblastoma and excluded 41. Of the 67 patients included in the analysis, 23 had mFI=0, 21 mFI=1, 15 mFI=22, and 6 had mFI >2. There was no significant difference in age (p=0.43) or KPS (p=0.25) between mFI2 vs. >2, nor in % biopsy (43% vs 33%, p=0.21). Patients with mFI>2 had shorter OS (20.8 18.4 mo vs 8.5 5.8 mo, p=0.01), shorter PFS6 (56% vs 27%, p=0.03), and OS at 24 months (28% vs 0, p<0.001). The more-frail patients were less likely to complete radiation treatment completion (p=0.06), more likely to have a lower radiation dose (p=0.07), and less likely to complete chemotherapy (p=0.33), though not statistically significance. They had longer hospital stay (p=0.04) and trended toward more rehospitalizations (p=0.08).
Conclusions: Frailty is an underused metric for risk assessment in geriatric patients with glioblastoma and may be a useful tool in addition to age and performance status to help guide shared decision making in this population.
Hemolytic Anemia as a Harbinger of Lymphoma
Megan Emmich, DO, Victoria Forbes, MD, Ritika Vankina, MD
UCONN Health, Carole and Ray Neag Comprehensive Cancer Center
Background: Hemolytic anemia produces variable degrees of clinical signs and symptoms and can co-exist with hematologic malignancies.
Methods: We describe a patient with hemolytic anemia preceding a diagnosis of lymphoma.
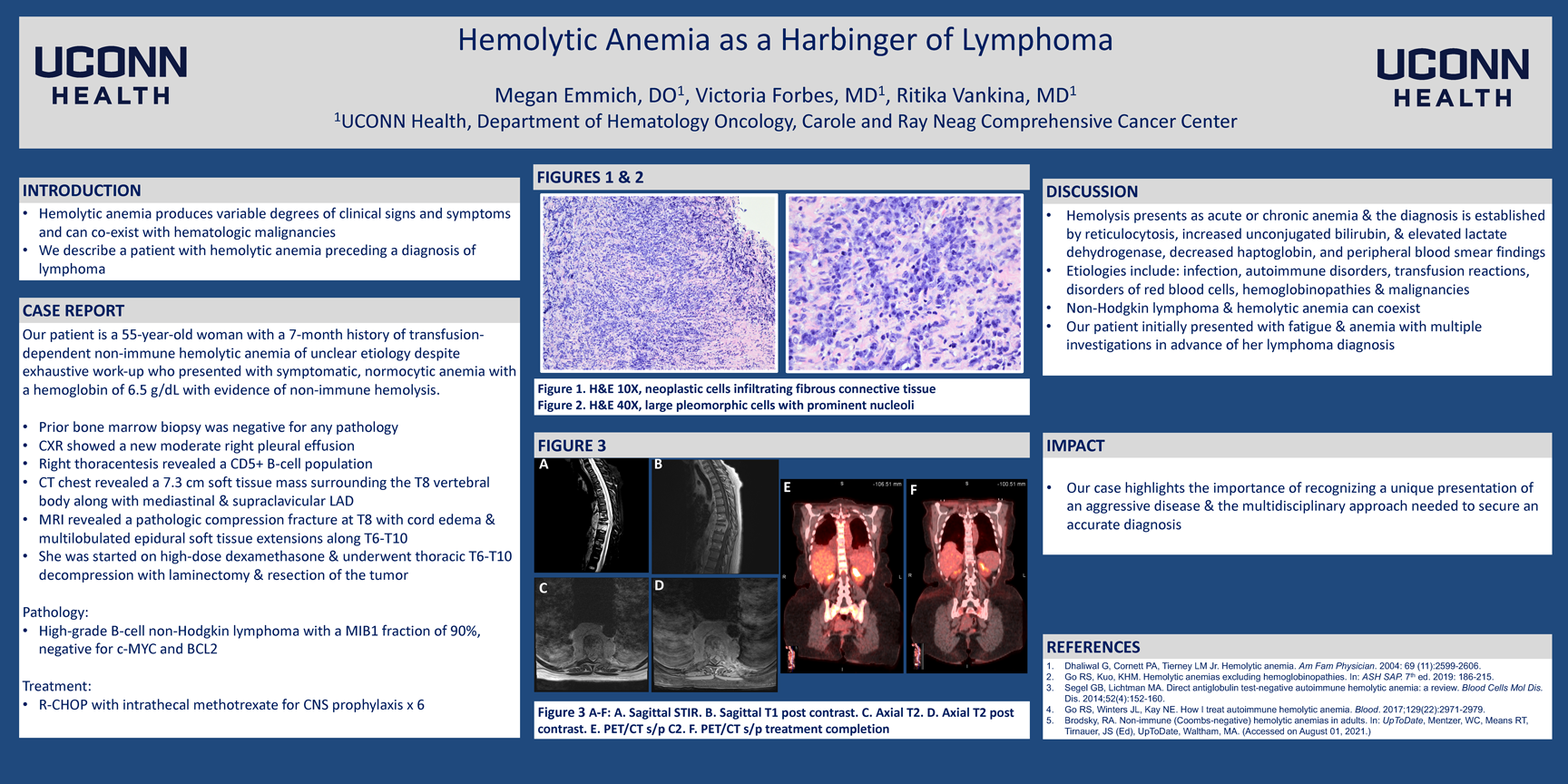
Results: Our patient is a 55-year-old woman with a 7-month history of transfusion-dependent non-immune hemolytic anemia of unclear cause despite exhaustive work-up who presented with symptomatic, normocytic anemia of 6.5 g/dL with evidence of non-immune hemolysis. A prior bone marrow was negative for any pathology. A chest x-ray showed a new moderate right pleural effusion. Right thoracentesis revealed a CD5+ B-cell population and CT chest revealed a 7.3 cm soft tissue mass surrounding the T8 vertebral body along with mediastinal and supraclavicular lymphadenopathy. MRI revealed a pathologic compression fracture at T8 with secondary cord edema and multilobulated epidural soft tissue extensions along T6 through T10. She was started on high-dose dexamethasone and underwent thoracic T6-T10 decompression with laminectomy and resection of the tumor. Pathology revealed a high-grade B-cell non-Hodgkin lymphoma with a MIB1 fraction of 90% and a negative c-MYC. She initiated R-CHOP therapy with intrathecal methotrexate for CNS prophylaxis.
Conclusions: Non-Hodgkin lymphoma and hemolytic anemia can coexist. The patient initially presented with fatigue and anemia and had multiple investigations before a diagnosis of lymphoma was made. This case highlights the importance of recognizing a unique presentation of an aggressive disease and the multidisciplinary approach needed to secure an accurate diagnosis.
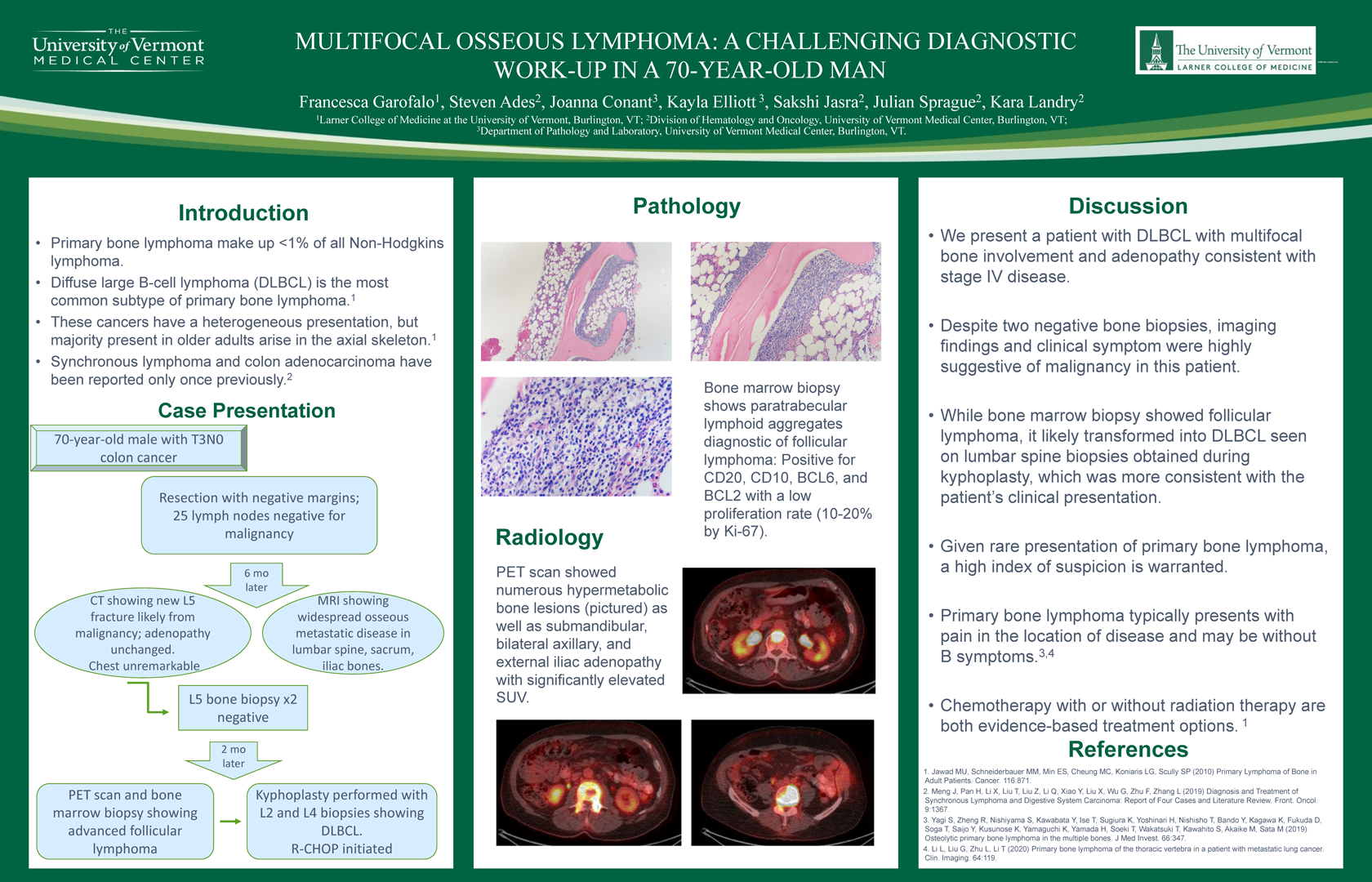
Multifocal osseous lymphoma: A challenging diagnostic work-up in a 70-year-old man
Francesca Garofalo1, Steven Ades2, Joanna Conant3, Kayla Elliott3, Sakshi Jasra2, Julian Sprague2, Kara Landry2
1 Larner College of Medicine at the University of Vermont, 2 Division of Hematology and Oncology, University of Vermont Medical Center, 3 Department of Pathology and Laboratory, University of Vermont Medical CenterBackground: Primary malignant bone lymphomas are rare, making up <1% of all non-Hodgkin’s lymphoma, and can vary in presentation. Diffuse large B-cell lymphoma is the most common subtype.
Results: We present a case of a 70-year-old male with a recent diagnosis of T3N0 colon cancer status post resection with negative margins and 25 lymph nodes negative for malignancy. A few slightly enlarged retroperitoneal lymph nodes (<11 mm) were noted on imaging at time of diagnosis but not amenable to biopsy. CT scan six months later showed new L5 vertebral fracture concerning for malignancy. Adenopathy was unchanged. CT chest was unremarkable. MRI showed widespread osseous metastatic disease in the lumbar spine, sacrum, and iliac bones. Bone biopsy of the L5 lesion was performed twice and was negative for malignancy. Due to worsening back pain, repeat imaging was obtained within 2 months and showed progressive metastatic disease within the spine. Bone marrow biopsy revealed follicular lymphoma that was MYC, CD20, CD10, BCL2, and BCL6 positive (Ki67 <30%). PET scan showed numerous hypermetabolic bone lesions as well as submandibular, bilateral axillary, and external iliac adenopathy with significantly elevated SUV. Kyphoplasty with additional biopsies of L2 and L4 was performed and demonstrated DLBCL. Treatment with R-CHOP was initiated.
Conclusions: We present a patient with DLBCL with multifocal bone involvement and adenopathy consistent with stage IV disease. Establishing a timely diagnosis can be challenging. The clinical course of our patient fit with an aggressive disease process, which was only established after multiple bone biopsies.
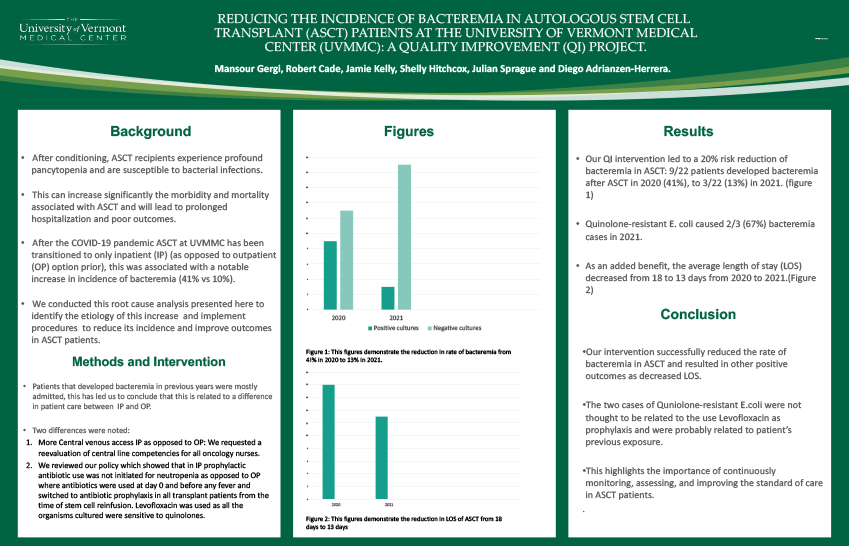
Reducing the incidence of bacteremia in autologous stem cell transplant (ASCT) patients at the University of Vermont Medical Center (UVMMC): A quality improvement (QI) project.
Mansour Gergi, MD, Jamie Kelly PA-C, Robert Cade PharmD, Shelly Hitchcox, Julian Sprague MD, Diego Adrianzen-Herrera MD
The University of Vermont Medical Center
Background: After conditioning, ASCT recipients experience profound pancytopenia and are susceptible to bacterial infections.
Methods: In 2020, we noted an increase in the rate of bacteremia among ASCT recipients compared to prior years at UVMMC (41% vs 10%). Due to COVID-19, all transplants were transitioned to inpatient (IP) since lodging was not available (historically, 60% ASCT are outpatient [OP]). Patients that developed bacteremia before 2020 were mostly IP, explaining the 2020 increase. More frequent central line manipulation in admitted patients compared to OP was identified. We requested a reevaluation of central line competencies for all oncology nurses and reviewed our policy, which did not mandate prophylactic antibiotics for admitted patients but did for OP, and switched to antibiotic prophylaxis in all transplant patients from the time of stem cell reinfusion. Levofloxacin was used as all the organisms cultured were sensitive to quinolones.
Results: Our QI intervention led to a 20% risk reduction of bacteremia in ASCT: 9/22 patients developed bacteremia after ASCT in 2020 (41%), while only 2/11 patients (22%) have developed bacteremia in 2021 so far, following a plan-do-study-act cycle. Quinolone-resistant E. coli caused 2/3 (67%) bacteremia cases in 2021. As an added benefit, the average length of stay (LOS) decreased from 18 to 13 days from 2020 to 2021.
Conclusions: Our intervention successfully reduced the rate of bacteremia in ASCT and resulted in other positive outcomes as decreased LOS. This highlights the importance of continuously monitoring, assessing, and improving the standard of care in ASCT patients.
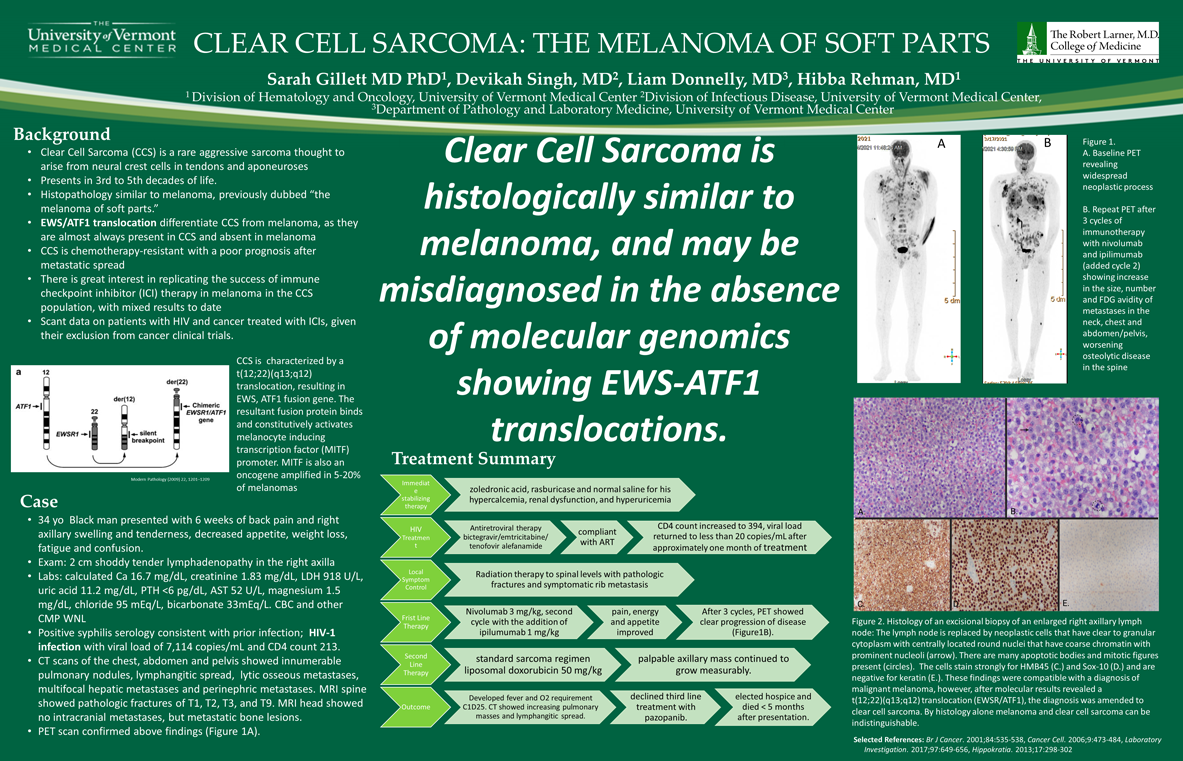
Clear cell sarcoma, the melanoma of soft parts
Sarah Gillett, MD, PhD1, Devika Singh, MD, MPH2, Liam Donnelly, MD3, Hibba Rehman, MD1
1 Division of Hematology and Oncology, University of Vermont Medical Center, 2 Division of Infectious Disease, University of Vermont Medical Center, 3 Department of Pathology and Laboratory Medicine, University of Vermont Medical CenterBackground: Clear cell sarcoma (CCS) is a rare aggressive sarcoma morphologically and histologically similar to melanoma, but may be differentiated by EWS-ATF1 translocations. Immune checkpoint inhibitor (ICI) may be a reasonable investigational therapy. There are scant data on patients with HIV and cancer treated with ICIs, given their exclusion from cancer clinical trials.
Methods: We report a case of clear cell sarcoma in a patient with HIV treated with ICI, palliative radiation, and chemotherapy.
Results: A 34 year-old male presented with 6 weeks of back pain and right axillary swelling. CT scans showed extensive malignancy with nodal, pulmonary, osseous, hepatic, and perinephric metastases. He was also found to have HIV-1 with a viral load of 7,114 copies/mL and a CD4 count of 213. Excisional biopsy of an axillary lymph node showed a metastatic poorly differentiated neoplasm consistent with malignant melanoma. Initial therapy consisted of palliative radiation, ipilimumuab and nivolumab for presumed melanoma and antiretroviral therapy for HIV. Genomics later revealed a EWSR1-ATF1 translocation, and his diagnosis was amended to clear cell sarcoma. Despite initial improvement in performance status and pain, after 3 cycles of immunotherapy, PET scan showed clear progression of disease. He started liposomal doxorubicin but his disease quickly progressed and he transitioned to hospice care and died less than 5 months after presentation.
Conclusions: CCS may be differentiated from melanoma by presence of EWS-ATF translocations. We report administration of ICI to a patient with CCS and HIV with initial symptomatic improvement but without long lasting control.
Compassion Fatigue in Nursing
Katie Hall, BSN, RN, OCN, ONN-CG
MSN-CNL UNH graduate programIntroduction/Background: Compassion fatigue is characterized by physical, emotional, social, spiritual, and professional implications. Nurses are especially vulnerable to developing compassion fatigue by nature of the enormous physical and emotional demands of the profession. To prevent compassion fatigue, the literature supports the importance of nurse recognition and being proactive. Does routine self-care/mindfulness practices reduce the prevalence of nursing related compassion fatigue, when compared to nurses who do not incorporate these practices?
Methods of Data Collection: To gain an understanding of the defining characteristics of compassion fatigue and effective strategies to mitigate the prevalence, a variety of databases were queried with the following search terms: compassion fatigue, nursing, oncology, mindfulness, and self-care, self-compassion, compassion satisfaction and resilience.
Practice implications: As the antidote to compassion fatigue, self- compassion is essential for nurses. Nurses can build resilience through self-compassion, in an effort to mitigate the effects of compassion fatigue. To promote a culture of awareness around compassion fatigue, it will be imperative nurse well-being and compassion satisfaction become a prominent focus in future nursing research and education. This approach will enhance knowledge, spread awareness, and reinforce preventative measures to build resilience for long, successful nursing
Data Summary: The literature and research supports self-care strategies and mindfulness-based interventions to be advantageous to nursing practice, ultimately leading to enhanced resilience and decreased burnout/compassion fatigue.
- Self-compassion and self-awareness are considered crucial components to cultivate compassion satisfaction and emotional wellbeing
- Awareness and early recognition are paramount to mitigate the effects of compassion fatigue
- Utilization of Professional Quality of Life (ProQOL) tool is recommended to measure compassion fatigue risk
Resources: https://www.coloradonursingcenter.org/ripple-effect-learning-sessions/
Outdoor Nourishing Activities Checklist
The Woe of the Warburg Effect
Emma L. Hanlon BS1, Yenong Cao MD PhD2, Margaret C. Liu MD2, York Chen MD2, Muhammad Z. Afzal MD MS3, Christi A. Hayes MD3, John M. Hill, Jr MD3
1 Geisel School of Medicine at Dartmouth, 2 Department of Internal Medicine, Dartmouth Hitchcock Medical Center,3 Division of Hematology and Oncology, Norris Cotton Cancer CenterBackground: The Warburg effect of high glucose uptake and lactate release by tumor is a rare condition potentially accompanying aggressive malignancies, and it carries a poor prognosis. This case demonstrates the importance of its prompt recognition, aggressive support and timely chemotherapeutic management.
Methods: A 76-year-old woman presented with the subacute onset of fatigue and weight loss. Initial labs included a leukocytosis to 24.2 x103/mcL, glucose of 9 mg/dL, pH 6.93, lactate of 19 units/L, and transaminitis. CT scan showed diffuse adenopathy, and she was ultimately diagnosed with aggressive lymphoma. Her lactic acidosis and severe hypoglycemia were felt consistent with the Warburg effect.
Results: While metabolic derangements improved with sodium bicarbonate infusion, lactate elevation persisted, prompting R-CHOP initiation prior to confirmation of the lymphoma subtype. Despite initial response to chemotherapy and lactic acidosis resolution, her course was subsequently complicated by renal failure, requiring CVVH treatment, then neutropenia, DIC and vancomycin resistant Enterococcus faecium sepsis, prompting transition to comfort care.
Conclusions: The effect of tumor anaerobic metabolism under aerobic conditions, characteristic of the Warburg effect, is felt to involve increased tumor production of lactate and decreased hepatic clearance. Optimal treatment of lactic acidosis in this setting includes a combination of bicarbonate administration and hemodialysis, with chemotherapeutic targeting of the underlying malignancy being the definitive treatment of choice. In this case, early chemotherapy initiation, sodium bicarbonate, and CVVH use to successfully resolve lactic acidosis, yet the patient’s ultimate demise, highlights the poor prognostic outlook forecasted by the Warburg effect.
References:
- Chan FH, Carl D, Lyckholm LJ. Severe Lactic Acidosis in a Patient with B-Cell Lymphoma: A Case Report and Review of the Literature. Case Rep Med. 2009;2009:1–6.
- Glasheen, Jeffrey, and Mathew Sorensen. "Burkitt's lymphoma presenting with lactic acidosis and hypoglycemia-a case presentation." Leukemia & lymphoma 46, no. 2 (2005): 281-283.
- Kim HJ, Son YK, An WS. Effect of sodium bicarbonate administration on mortality in patients with lactic acidosis: a retrospective analysis. PloS One. 2013;8(6):e65283.
- Keller G, Cour M, Hernu R, Illinger J, Robert D, Argaud L. Management of metformin-associated lactic acidosis by continuous renal replacement therapy. PloS One. 2011;6(8):e23200.
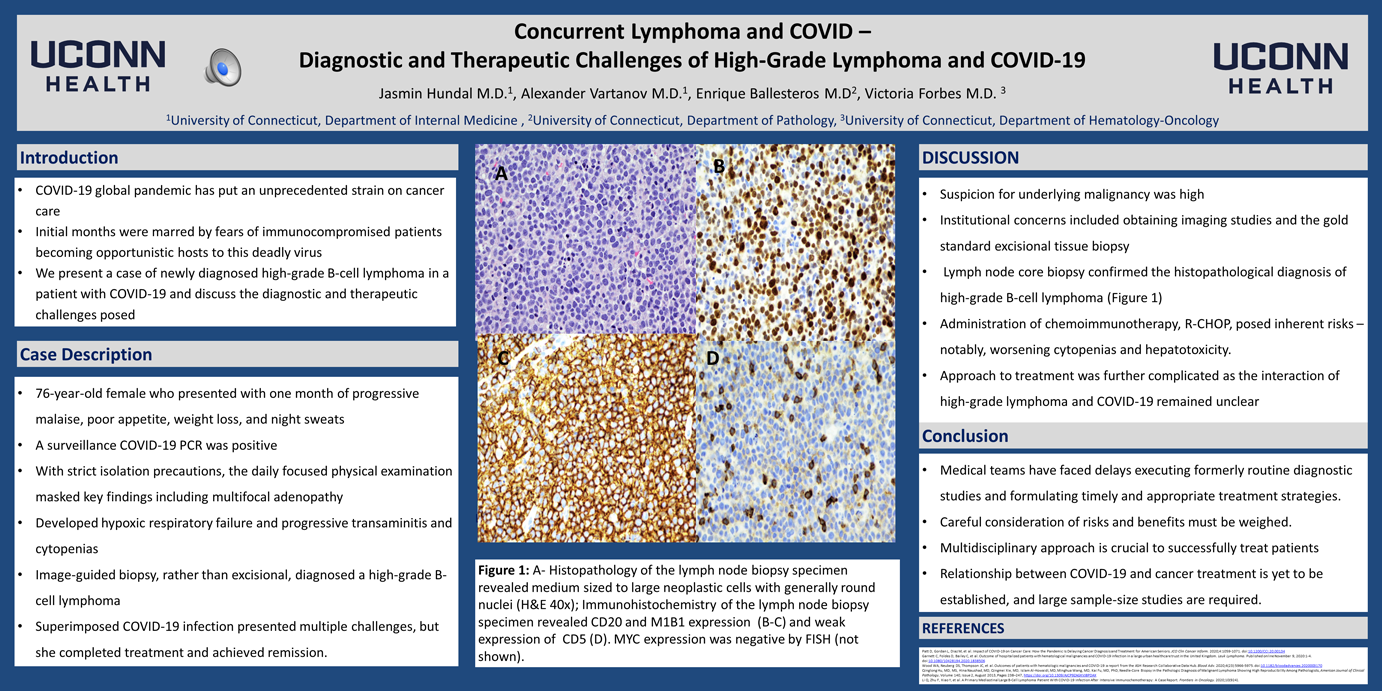
Concurrent Lymphoma and COVID - Diagnostic and Therapeutic Challenges of High-Grade Lymphoma and COVID-19
Jasmin Hundal M.D., Alexander Vartanov M.D., Enrique Ballesteros M.D., Victoria Forbes M.D.
University of Connecticut Health CenterBackground: The COVID-19 global pandemic has put an unprecedented strain on cancer care. The initial months were marred by fears of immunocompromised patients becoming opportunistic hosts to this deadly virus. We present a case of newly diagnosed high-grade B-cell lymphoma in a patient with COVID-19 and discuss the diagnostic and therapeutic challenges posed.
Methods: Our patient is a 76-year-old female who presented with one month of progressive malaise, poor appetite, weight loss, and night sweats. A surveillance COVID-19 PCR was positive. With strict isolation precautions, the daily focused physical examination masked key findings including multifocal adenopathy. She developed hypoxic respiratory failure and progressive transaminitis and cytopenias. Image-guided biopsy, rather than excisional, diagnosed a high-grade B-cell lymphoma. Superimposed COVID-19 infection presented multiple challenges, but she completed treatment and achieved remission.
Results: Suspicion for underlying malignancy was high. Institutional concerns included obtaining imaging studies and the gold standard excisional tissue biopsy while maintaining acceptable staff exposure. Fortunately, a lymph node core biopsy confirmed the histopathological diagnosis of high-grade B-cell lymphoma (Figure 1). The administration of chemoimmunotherapy, R-CHOP, posed inherent risks – notably, worsening cytopenias and hepatotoxicity. The approach to treatment was further complicated as the interaction of high-grade lymphoma and COVID-19 remained unclear.
Conclusions: Medical teams have faced delays executing formerly routine diagnostic studies and formulating timely and appropriate treatment strategies. Careful consideration of risks and benefits must be weighed. A multidisciplinary approach is crucial to successfully treat patients. The relationship between COVID-19 and cancer treatment is yet to be established, and large sample-size studies are required.
Hemophagocytic Lymphohistiocystosis (HLH) secondary to Immune Checkpoint Inhibitor Therapy for Metastatic Lung Adenocarcinoma
Samer Ibrahim, DO1, Mark Hanna2, Muhammed Zubair Afzal1, Konstantin H. Dragnev, MD1, Swaroopa Yerrabothala, MD1
1 Norris Cotton Cancer Center at Dartmouth-Hitchcock Medical Center, 2 New York Institute of Technology College of Osteopathic MedicineBackground: HLH is a disorder of uncontrolled activation of the immune system leading to life-threatening complications. Acquired HLH due to infections, malignancy, and rheumatologic disorders have been increasingly reported in recent years.
Methods: We present a case of a 62-year-old female with metastatic adenocarcinoma of the lung who was initiated on carboplatin, pemetrexed, and the immune checkpoint inhibitor; pembrolizumab. She initially experienced thrombocytopenia, which was thought to be due to the cytotoxic chemotherapy, ultimately leading to treatment delays and dose reduction. Therapy with pembrolizumab alone was attempted but her thrombocytopenia sharply worsened with a new onset of anemia. A bone marrow biopsy showed significant hemophagocytosis and focal emperiopolesis.
Confirmation testing for HLH showed a soluble IL-2R greater of 2,870 U/mL, CXCL9 assay of 151 pg/mL (< 121) and a low NK cell activity. Ferritin peaked at 17,972.
Results: We started treatment with etoposide and steroids. However, the patient’s liver function acutely worsened following the first cycle due to the HLH and her history of liver metastasis. A second dose-reduced cycle was attempted but the patient continued to decline and opted towards comfort-oriented care.
Conclusions: As new indications for immune checkpoint inhibitors are approved, we also discover previously unknown adverse events of these treatments. Our patient was noted to have cytopenias after treatment with pembrolizumab and was diagnosed with HLH. Her clinical picture quickly declined and she transitioned to comfort-oriented care despite attempts at treatment.
Mast Cell Leukemia after t(8,21)AML ; a challenge in diagnosis and treatment
Lakshmi Kolandra1, Dr. William Baker1, Dr. Swarup Kumar1, Dr. Meghana Singh1, Dr. Adriana Kahn2
1 UConn Health, 2 Yale New Haven HealthBackground: Mast cell leukemia (MCL) is a form of systemic mastocytosis (SM) that occurs in <1% of cases of mast cell disorders. Given the rarity of this disease, treatment for MCL is based on data derived from treatment of SM.
Methods: We present the case of a 50 year-old male with history of acute myeloid leukemia (AML). Cytogenetics showed t(8;21)(q22;q22) with RUNX1-RUNX1T1 rearrangement. He underwent induction chemotherapy with cytarabine and idarubicin and bone marrow biopsy (BMB) at the end demonstrating complete remission with the absence of the RUNX1-RUNX1T1. He then underwent consolidation with high-dose cytarabine. A few weeks after achieving remission, he presented to the hospital with fevers, diarrhea and thrombocytopenia. Repeat BMB now showed a dense CD117+/CD25+ monocytoid mast cell infiltrate suggestive of frank MCL. Mutations of the c-kit gene were not present. The leukemic myeloblasts in BMB at presentation were not identified, but RUNX1T1-RUNX-1 fusion was found in 4% of cells analyzed by FISH. These pathologic findings with elevated tryptase levels (799 ug/L) and osteolytic lesions on imaging led to the diagnosis of MCL. He was treated with a 5 day course of cladribine and plan to begin midostaurin. Unfortunately, his condition deteriorated and he expired.
Results: As the incidence of MCL is very low, the diagnosis and treatment of this disease remains a clinical challenge. Even rarer is the occurrence of MCL after t(8,21)AML of which only a handful of cases have been reported.
Conclusions: Further research may ultimately lead to heightened awareness and improved prognosis of this disease.
Uptake of prevention strategies by women at high risk of developing breast cancer
The University of Vermont Medical Center
Background: Uptake of preventative treatment (chemoprevention, prophylactic surgery) among women at risk for breast cancer is low. We evaluated breast cancer risk factors as predictors of preventative treatment (PT).
Methods: We analyzed a prospective cohort of women at risk for breast cancer enrolled in an IRB-approved registry between 2003-2020 at the University of Vermont. We examined the association between breast cancer risk factors (age, BMI, family history, genetic predisposition, benign breast disease and modeled risk) and uptake of PT. We used logistic regression to estimate odds ratios (OR).
Results: 509 women followed for median of 12.5 years were included. Mean age was 45 years (range 19.0 – 78.0). 78% had a family history of breast cancer, 60% had a high modeled breast cancer risk, and 14% had a history of benign breast disease. 8% of women had confirmed genetic risk. 18% underwent PT. PT uptake was highest among women aged 60-69 years; the ORadj was 6.48 relative to women aged <40 (95% CI 2.25–18.67, p <0.001). Having a genetic predisposition (ORadj 8.49, 95% CI 2.30–31.35, p=0.001) and history of benign breast disease (ORadj 3.98, 95% CI 1.81–8.72, p<0.001) were also associated with uptake of prevention strategies.
Conclusions: Like other studies, we found a low uptake of PT (18%) among women at risk for developing breast cancer. Age, genetic predisposition, and benign breast disease were associated with uptake of PT. Understanding barriers to uptake among women in this cohort may identify opportunities for development of interventions to improved uptake.
Leveraging the Delphi Method to develop an evidence-based prehabilitation regimen
Laura Nicolais1, Abdimajid Mohamed, BS2, Amy Litterini PT, DPT1, Timothy Fitzgerald, MD, FACS1
1 Maine Medical Center, 2 Tufts Medical UniversityBackground: Frail surgical oncology patients have increased mortality, complications, and loss of independence. Prehabilitation may improve outcomes; however, research is inconsistent, possibly due to a lack of empirically derived regimens.
Methods: The Delphi Method was implemented with a panel of seven academic physical therapists. A series of three questionnaires (Q) were administered: Q1) open-ended to identify possible interventions, Q2) rating the importance of interventions identified in Q1, Q3) rating the importance of the top four interventions identified by group consensus in Q2.
Results: In Q1 respondents recommended light cardiovascular (100%), strength (83.3%), functional training (33.3%), fall prevention (50%), stretching (16.7%), and pelvic girdle (16.7%). Q2 and Q3 utilized 6-point rating scales. In Q2 the mean of ratings were as follows: cardiovascular (2.4), strength (2.9), functional training (3), fall prevention (3.4), stretching (4.7), and pelvic girdle exercise (4.6). Q3 resulted in a group consensus determining the ideal prehabiliation program includes light cardiovascular (2.14), strength training (2.71), and functional training (2.14).
Follow-up questions in Q2 suggest that cardiovascular training should occur for 30-minutes daily with an initial intensity of 3-5/10 [Borg RPE Scale] and a maximum of 6-7/10. Additionally, patients should start with 1 set of 10 repetitions, increasing to a maximum of 3 sets of 15 repetitions.
Conclusions: The Delphi Method was implemented to develop an empirically derived prehabilitation program. Future research will examine the effectiveness of the three identified interventions for mitigating poor surgical outcomes for oncology patients.
Development of a Comprehensive Web-based Prehabilitation Program for Gastrointestinal Cancer Survivors
Eric Norman1, Maryam Nahidian1, Amy Litterini2, Dr. Timothy Fitzgerald3
1 University of New England Doctorate of Physical Therapy Student, 2 MaineHealth Cancer Care Network, 3 Chief of Surgical Oncology, Maine Medical CenterBackground: Individuals with frailty who undergo surgical procedures for gastrointestinal cancers are more likely to experience post-surgical complications, have a higher readmission rate, are more likely to be discharged to skilled care, and have an over four-fold risk of mortality. Developing targeted patient education tools and resources may support, and reduce complications for, individuals with frailty undergoing and recovering from major abdominal surgery.
Methods: Program development began via the Delphi Method, including three survey iterations distributed to top experts in the field of Oncologic Physical Therapy. Results achieved expert consensus for exercise parameters. Brief, simple to understand exercise videos and instructions were created for patients to perform independently preoperatively to increase their physical capabilities at home. Other key stakeholders including the surgical oncologist, pre-operative admissions staff, surgical care clinical manager, surgical oncology navigators, physical therapists and education specialists created the complete prehabilitation program curriculum within the Thinkific on-line education platform with input from cancer survivors.
Results: The final curriculum included: prehabilitation instructions for pre-surgical preparation, therapeutic exercise, and nutrition optimization; tobacco cessation resources; instruction in stress management and relaxation techniques; and postoperative delirium reduction strategies and incision care. Promotional materials were created to launch the pilot program within the surgical oncology service line.
Conclusions: This process improvement project proved to be feasible and appropriate for this application as a new oncology service. Future directions include program implementation for gastrointestinal cancer survivors with the intention for outcomes data collection. Further implementation is planned for cancer survivors with different diagnoses facing surgery.
USE OF A PATIENT SURVEY TO MONITOR STATIN-PACLITAXEL INTERACTION
Michael Pelchat, RPh
NH Oncology-Hematology PA
Paclitaxel binds to cellular microtubules rendering them non-functional ultimately leading to cell death. This action on microtubules also impacts neurons which rely on microtubules for nutrient and growth factor transport. Inhibiting this function is thought to be one of the causes of paclitaxel neuropathy. Paclitaxel is removed from the neuron by p-glycoprotein and it has been shown that inhibiting p-glycoprotein with drugs such as statins can enhance paclitaxel neurotoxicity.
We developed a survey to detect the statin-paclitaxel interaction. The survey quantified toxicity symptoms and their impact on daily activities. Surveys were evaluated by a pharmacist and if toxicity was severe enough to warrant a dose reduction, provider was asked to d/c the statin. Patients completed the survey prior to each paclitaxel treatment.
173 paclitaxel patients were reviewed. 42 patients taking statins were surveyed, 131 not on statins had a chart review. For patients not on a statin, 66% had documented toxicity symptoms while 34% did not. Survey results on the 42 patients taking a statin showed 81% with symptoms and 19% without. Seven patients had severe symptoms and the statin was d/c’d. Of these, 2 had some improvement, 1 had no change and 4 had worsening symptoms.
The addition of a statin increases the incidence of toxicity. Though not showing significance (p= <.06), the trend was toward an increased risk. Stopping the statin had no apparent impact in reducing toxicity. Perhaps the bar was set too high and stopping the statin as soon as symptoms occurred should be considered.
VTE Risk Assessment and Prevention in Cancer Patients in the Community Oncology Setting*
Yoni Resnick1, Dr. Chris Holmes2, Dr. Steven Ades2, Melissa Timberlake, NP1, Jacob Barker, PharmD2, Dr. John Winters1, Karen Libby, RN2, Dr. Christian Thomas1, Emily Parenteau, RN1
1 New England Cancer Specialists, 2 The University of Vermont Medical CenterBackground: National guidelines recommend VTE risk assessment in cancer outpatients and pharmacologic thromboprophylaxis in high risk patients. Despite validated risk stratification tools, rates of VTE prophylaxis in high-risk patients are low. A successful model for guideline implementation, VTEPACC, was recently reported in an academic setting in Vermont. The objective of this study was to determine the success of a VTE prevention model at New England Cancer Specialists (NECS), a community oncology practice.
Methods: VTEPACC at NECS is a multidisciplinary program involving EMR changes, provider education, and workflow meetings prior to implementation. Over a 6 month timeframe, we collected data on VTE education rates, risk assessment rates, accuracy of risk assessment, rate of VTE prophylaxis and side effects of VTE prophylaxis.
Results: 302 patients met inclusion criteria for VTE risk assessment. VTE risk education was provided to every patient. 52 patients (17.2%) were scored as high risk, of whom 14 (26.9%) were initiated on thromboprophylaxis. 5 patients (35%) discontinued anticoagulation due to mild bleeding. Barriers to implementation included geographic diversity of a community practice with multiple clinic sites, nuances associated with the Khorana score, EMR optimizations, higher than expected miscalculations of the risk score, and economic issues.
Conclusions: Involving a multidisciplinary team, implementation of a VTEPACC model was successful at NECS. VTE education rates skyrocketed and nearly all patients had VTE risk assessed. Over time, solutions to barriers were identified. Our experience demonstrates that implementation of a VTE risk assessment program is an achievable endeavor that can be mimicked at other community oncology practices.
Adrenal, Small Intestine and Bone Metastasis from Sarcomatoid Carcinoma of the Pancreas: A Rare Case with Long-term Survival
Ivy Riano, M.D, Kathryn Hourdequin, M.D.
Norris Cotton Cancer Center, Dartmouth-Hitchcock Medical CenterBackground: Sarcomatoid carcinoma of the pancreas (SCP) is an extremely rare malignancy, with only a few cases reported in the literature. SCP exhibits high rates of local recurrence and distant metastasis. Thus, the prognosis is poor with a median overall survival of 8 months.
Methods: Herein, we report a case of SCP with 10-year survival after the surgery.
Results: A 54-year-old Caucasian male presented with epigastric pain in February/2011. A CT scan showed a hypodense 22 x 23 x 24 mm mass within the pancreatic tail. FNA biopsy revealed a poorly differentiated malignant neoplasm with sarcomatoid features and extensive necrosis. CA 19-9 was <5.0u/mL. He received neoadjuvant chemotherapy and underwent distal pancreatectomy. Subsequently, the patient was found with bone and mediastinum metastasis and was treated with chemo-radiation. In February/2017, a CT scan showed a 12 mm right adrenal nodule suspicious for metastasis; the patient underwent right adrenalectomy (1.7 cm). Pathology revealed high-grade undifferentiated sarcomatoid neoplasm. He was treated with salvage radiotherapy. In March/2021, a CT scan revealed lobular soft tissue mass within the mid jejunum with increased activity in PET/CT. The patient underwent laparoscopic-assisted small bowel resection of metastasis (5.4 cm). Pathology revealed sarcomatoid neoplasm. At 10-year after diagnosis, the patient is alive with recurrence to the left adrenal gland.
Conclusions: Our patient is one of the few cases with long survival reported in the literature. Owing to the rarity of the SCP, this report aims to provide a better understanding of the clinical-pathological features of this poorly characterized entity.
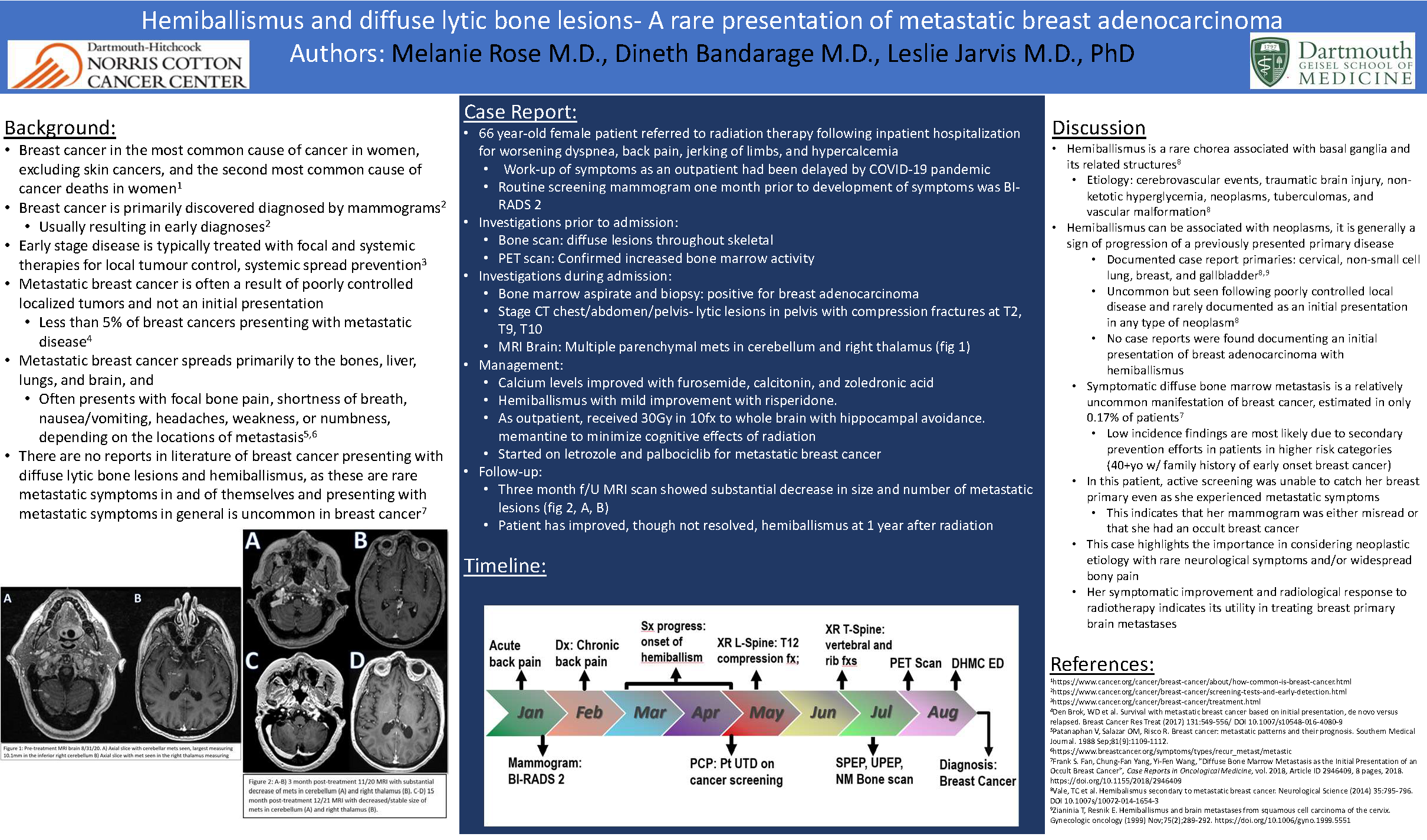
Hemiballismus and diffuse lytic bone lesions- A rare presentation of metastatic breast adenocarcinoma
Melanie Rose, Lesley Jarvis
Dartmouth-Hitchcock Medical CenterBackground: Breast cancer is a common cause of cancer in women and mostly found in early stages given regular surveillance. Metastatic presentation is uncommon, brain mets even less so. Common symptoms of brain metastases include headaches, nausea, and sensation/strength changes. Hemiballismus would be considered a rare symptom.
Case Presentation: 66yo female with no significant past medical history and recent mammogram with bi-rads2 experienced 8 months of bony pain and 3 months of new neurological symptoms, including hemiballismus. Previous work-up prior to presentation was limited and delayed due to the COVID19 pandemic. Outpatient work-up had included a thoracic radiograph, nuclear medicine bone scan, and PET scan, which had confirmed diffuse bony lesions. During her hospitalization, bone biopsy confirmed primary adenocarcinoma of the breast, with discrete 2cm breast mass palpated on the left areolar border. Calcium was found to be elevated, which was presumed to be the cause of her neurological symptoms, but on resolution of the hypercalcemia she continued to experience hemiballismus. MRI brain confirmed multiple brain metastasis and on discharge she was seen by radiation oncology where she received whole brain radiation therapy in 30Gy in 10fx. On subsequent follow-ups imaging revealed resolution of brain metastases and markedly improved hemiballismus, although no complete resolution.
Conclusions: This case highlights the importance in investigating and considering neoplastic etiology with rare neurological symptoms and/or widespread bony pain. Her symptomatic and radiological response to radiotherapy indicates its utility in treating breast primary brain metastases, and should be considered in similar cases as a viable and non-invasive treatment.
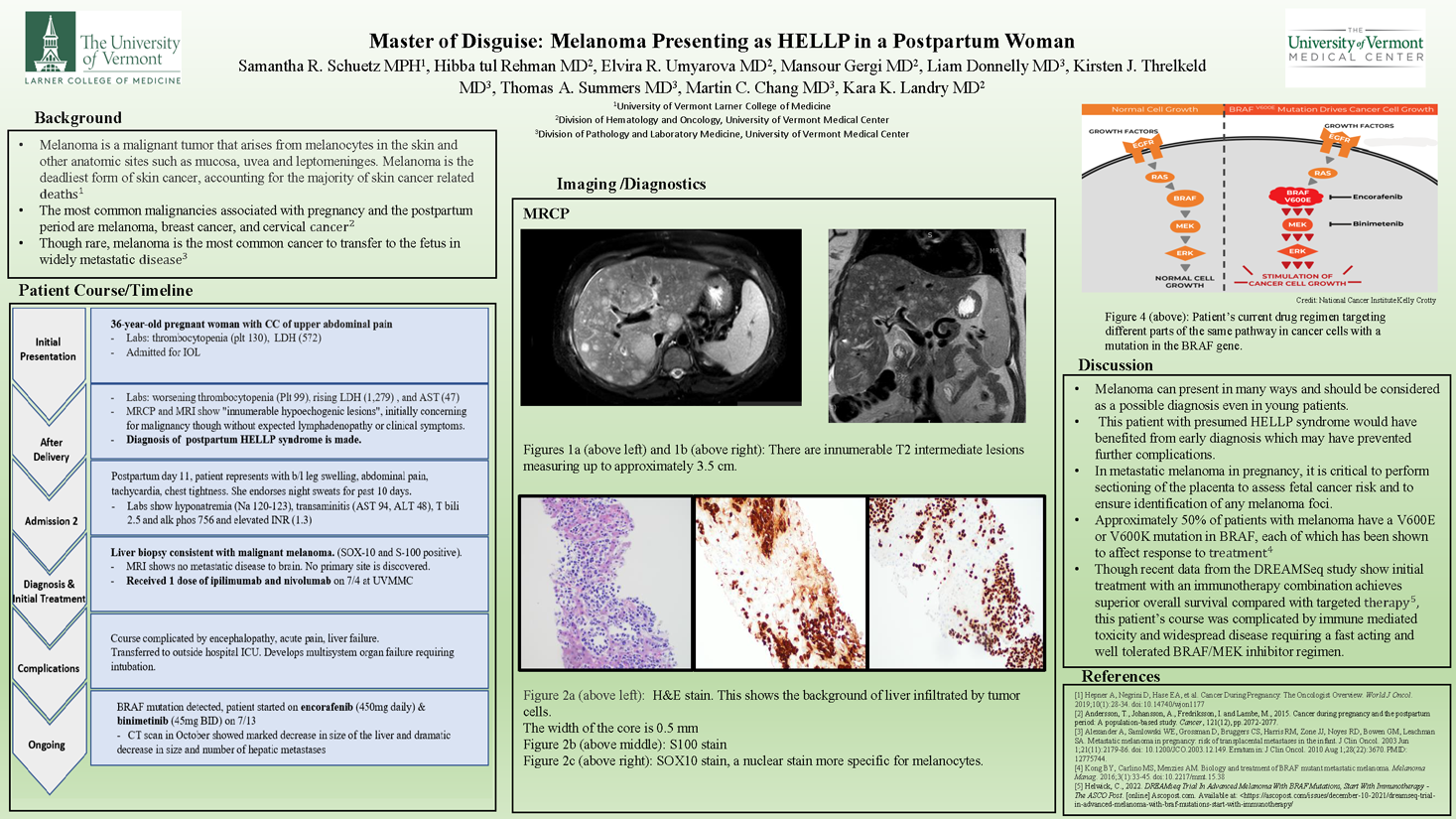
Master of disguise: melanoma presenting as HELLP in a postpartum woman
Samantha R. Schuetz, MPH, Hibba tul Rehman, Elvira R. Umyarova, Mansour Gergi, Liam Donnelly, Kirsten J. Threlkeld, Thomas A. Summers, Martin C. Chang, Kara K. Landry
The University of Vermont Medical CenterBackground: Melanoma is one of the most common cancers diagnosed during pregnancy, and though rare, is the most common cancer to transfer to the fetus in cases of widely metastatic disease.
Methods: Case Report
Results: We present a case of a 36-year-old woman who underwent induction of labor for abdominal pain, thrombocytopenia and elevated liver enzymes. Following delivery, the patient was clinically diagnosed with HELLP syndrome. After two ED visits for abdominal pain, dyspnea, tachycardia, and night sweats, she was re-admitted. Imaging showed multifocal liver lesions and left axillary adenopathy. Liver biopsy demonstrated metastatic melanoma (SOX-10 and S-100 positive). MRI brain revealed no evidence of metastases and dermatologic exam revealed no primary cutaneous lesion. The placenta was not available to assess for metastases. While awaiting BRAF testing, she received one cycle of nivolumab and ipilimumab. Her course was complicated by hyponatremia, metabolic encephalopathy, pseudocirrhosis and multiorgan failure requiring ICU and intubation. Testing revealed a BRAF activating mutation and binimetinib and encorafenib were started. She is currently extubated and clinically improving.
Conclusions: Melanoma can present in many ways and should be considered as a differential diagnosis even in young patients. Our patient with presumed HELLP syndrome could have benefited from early diagnosis which could have prevented further complications. In metastatic melanoma in pregnancy, it is critical to perform sectioning of the placenta to assess fetal cancer risk and to ensure identification of any melanoma foci.
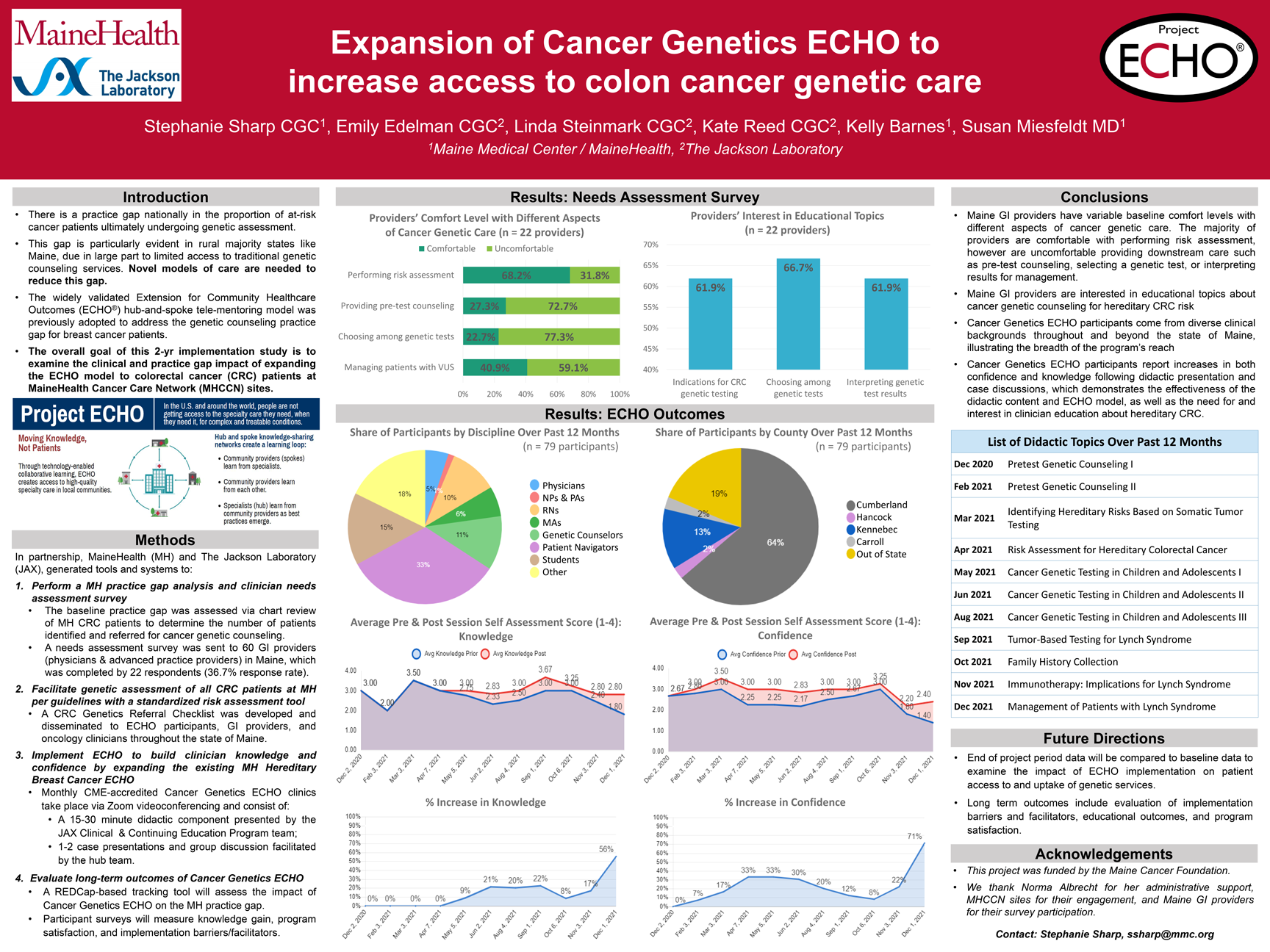
Expansion of Cancer Genetics ECHO to increase access to colorectal-related cancer genetic care
Stephanie Sharp, MS, CGC1, Emily Edelman, MS, CGC2, Kate Reed, MPH, ScM, CGC2, Linda Steinmark, MS, CGC2, Susan Miesfeldt, MD1
1 Maine Medical Center, 2 The Jackson LaboratoryBackground: There is a colorectal cancer (CRC) genetic counseling gap nationally, especially in rural states like Maine with limited access to these services, calling for novel care models.
Overall goal: To increase MaineHealth (MH) CRC patient access to cancer genetic services by building clinician capacity to i) identify at-risk patients and ii) facilitate provision of genetic counseling/testing through implementation of the Project ECHO® hub-and-spoke telementoring model.
Methods: In partnership, Maine Medical Center and The Jackson Laboratory generated tools and systems to i) perform a MH practice gap analysis and clinician needs assessment survey, ii) ensure genetic assessment of all CRC patients at MH per guidelines with a standardized risk assessment tool, and iii) implement ECHO to build clinician knowledge and confidence by expanding the existing MH hereditary breast cancer ECHO.
Results: Providers (n=22) had variable baseline comfort levels with different aspects of cancer genetic care. Most (68.2%) were comfortable performing point-of-care risk assessment, and uncomfortable providing pretest counseling (72.7%), choosing among genetic tests (77.3%) and managing patients with variants of uncertain significance (59.1%). Providers were most interested in education about choosing tests (66.7%), indications for genetic testing (61.9%) and interpreting test results (61.9%). Cancer Genetics ECHO participation and engagement is tracked for educational outcomes.
Conclusions: Long-term outcomes include Cancer Genetics ECHO impact on the MH practice gap; surveys will measure knowledge gain, implementation barriers/facilitators, and program satisfaction. This work promises to serve as a model for other NNECOS state-affiliates and nationally.
Notes: Acknowledgement: Work funded through the Maine Cancer Foundation
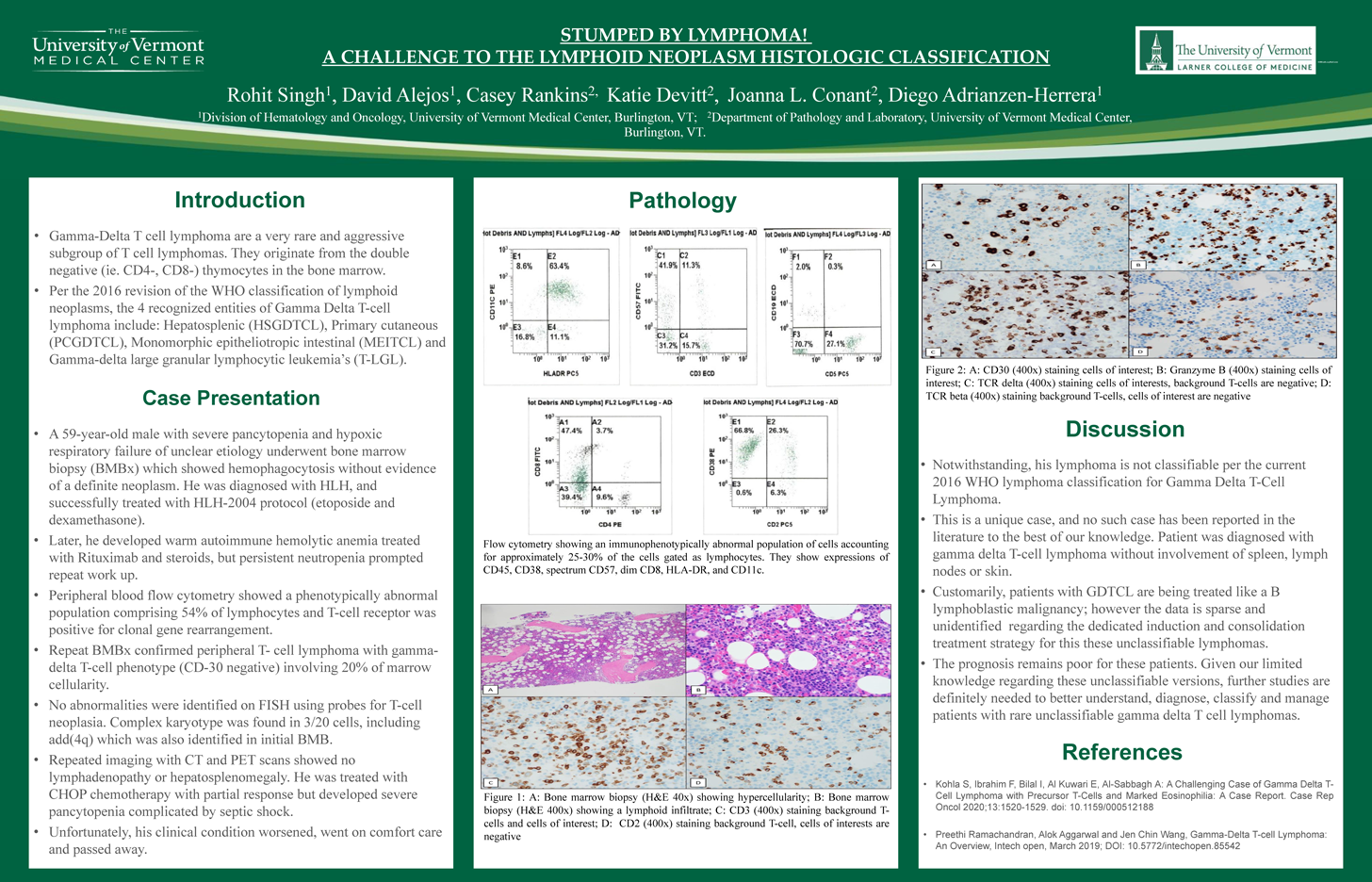
Stumped by Lymphoma! A challenge to the Lymphoid Neoplasm Histologic Classification
Rohit Singh1, David Alejos MD1, Casey Rankins DO2, Joanna L. Conant MD2, Diego Adrianzen-Herrera MD1
1 Department of Hematology-Oncology, University of Vermont Medical Center, 2 Department of Pathology & Laboratory Medicine, University of Vermont Medical CenterBackground: In the current WHO classification of lymphoid neoplasms, two entities of gamma delta T-cell origins are recognized: Hepatosplenic T-cell lymphoma and primary cutaneous gamma delta T-cell lymphoma. Peripheral gamma delta T-cell lymphoma (PGD-TCL) isolated to blood and bone marrow is rare and an aggressive subgroup of T-cell lymphomas.
Methods: Case Report
Results: A 59-year-old male with pancytopenia and hypoxic respiratory failure underwent bone marrow biopsy (BMB) showing hemophagocytosis without evidence of malignancy. He was diagnosed with hemophagocytic lymphohistiocytosis (HLH) and successfully treated with HLH-2004 protocol (etoposide and dexamethasone). He subsequently developed warm autoimmune hemolytic anemia and was treated with Rituximab and steroids. Persistent neutropenia prompted repeat evaluation. Peripheral blood flow cytometry showed a phenotypically abnormal population comprising 54% of lymphocytes and T-cell receptor analysis was positive for clonal gene rearrangement. Repeat BMB confirmed involvement by CD30-positive PGD-TCL involving 20% of marrow cellularity. Complex karyotype was found in 3/20 cells, including add(4q) which was also identified in initial BMB. No abnormalities were identified by FISH. Repeated imaging with CT and PET scans showed no lymphadenopathy or hepatosplenomegaly. He was treated with CHOP chemotherapy with partial response but ultimately his clinical condition deteriorated and was transitioned to comfort care.
Conclusions: PGD-TCL isolated to blood and bone marrow, without nodal or extra-nodal involvement, is rare and does not fit into the current WHO classification of lymphoid neoplasms. Clinicopathologic characteristics may help recognition of this aggressive subtype. In this case, add(4q) may represent a cytogenetic marker.
Budgetary impact of COVID-19 on prescribing of pegfilgrastim and its biosimilar
Patrick Skeffington, PharmD, MHA, MSRA, Bethany Gomes, PharmD,Annmarie Santos, RPh, Janice DallaCosta, RN, OCN
Southcoast Center for Cancer CareBackground: Covid-19 has caused profound changes in our healthcare model and how we deliver care to cancer patients. During the darkest days of the pandemic, cancer centers were forced to keep patients safe by limiting their contact with people and keeping them out of the clinics and hospitals. Southcoast Centers for Cancer Care was one such center.
Methods:
- Usage of Pegfilgrastim (Onpro) was tracked during a six-month period of COVID-19
- Usage of the same preparation was tracked during the six-month period prior year
- Treatment plans were not rewritten for this switch
Results:
During the time frame October 2019 to March 2020 (Seventeen patients):
- Eleven patients started on treatment plans for which Pegfilgrastim was warranted (built with Udenyca) but were switched to Onpro
- Five patients had counts that plummeted, so support was warranted (patient specific)
- Six patients began therapy on a Friday
During the time frame October 2020 to March 2021 (Twenty-seven patients):
- Fourteen patients were started on treatment plans for which Pegfilgrastim was warranted (built with Udenycaall received Onpro
- Six patients had counts that plummeted, so support was warranted (patient specific), all of these used OnPro
- Ten patients began therapy on a Friday
Conclusions: Present costs of Onpro are $2390.00 and Udenyca $1,255.00, difference of $1,135.00. During the six-month sample period preceding COVID, 27 doses or $20,430 was spent on Onpro doses that were changed from Udenyca. During the COVID period, 108 doses or $112,395.00 was spent on Onpro that were purposefully changed from Udenyca.
Fighting the War against COVID-19: The administration of Bamlanivimab (BAM) or Bamlanivimab plus Etesevimab (BAM+E); a cooperative effort between a community cancer center and an urgent care (UC) facility
Patrick Skeffington, PharmD, MHA, MSRA, Robert Aisenberg MD Pulmonology, Janice DallaCosta RN Oncology Nurse Manager, Ian Donaghy RN, Dani Hackner MD Chief Clinical Officer, Kelly Houde RN CEN Practice Administrator, Kathy Moraes RN Director Medical Oncology and Annemarie Santos, RPh
Southcoast Center for Cancer CareBackground: Goal of Massachusetts DPH is to ensure equitable distribution of BAM to most vulnerable, at risk of poor outcomes and to communities with highest incidences of COVID-19. Hospitals should allocate doses in a manner consistent with this guidance:
- Patients who meet Emergency Use Authorization (EUA) criteria; lottery system used if supply is exceeded
- BAM was approved for the treatment of mild/moderate COVID-19 for those at risk of progressing to severe disease (revoked 4/16/2021)
- BAM+E was approved for the same patients and criteria (revoked 6/25/2021)
Methods:
- Patients identified based on algorithm using Social Vulnerability Index (SVI) and EUA criteria
- RNs screened cases for positive criteria using SVI
- Pulmonologists consented appropriate patients, ordered infusions, routed cases for scheduling within window of treatment
- Experienced nursing staff from Southcoast departments treated patients
- Oncology pharmacy reviewed, prepared and delivered the BAM or BAM+E infusions once patient assessed by RN
Results:
- 2.5 days; symptom onset to test
- 5 days; test to treatment
- 5.0% inpatient admissions within 14 days
- 11.5% ED/UC visits within 14 days
- 1.4% inpatient admissions in 28 days
- 5.0% ED/UC visits within 28 days
- Two deaths during analysis of all cases
Conclusions: Cooperative effort between Cancer Center and Urgent Care led to positive outcomes for COVID- 19 patients. Southcoast demonstrated a 5% hospital admission rate for COVID-19 patients in the MAB program versus a 26% admission rate overall for COVID-19 patients.
A case of Hereditary Coproporphyria with an acute porphyria attack triggered by rifampin
Anya Srikureja1, Emily R. Straley1, Rahul Anil2
1 Larner College of Medicine, 2 University of Vermont Medical CenterBackground: Hereditary Coproporphyria (HCP) is a rare autosomal dominant acute hepatic porphyria caused by a mutation in coproporphyrinogen oxidase, an enzyme in the heme synthesis pathway. The buildup of toxic intermediates leads to neurovisceral attacks that are precipitated by a variety different triggers. The low incidence and nonspecific symptoms of HCP can result in delays in diagnosis and treatment.
Methods: Case Report
Results: A 53 year old male with no significant past medical history presented with several days of nausea, vomiting, constipation and severe abdominal pain. History revealed that the patient had recently been started on a course of Rifampin for treatment of asymptomatic Bartonella. An extensive lab work-up over multiple admissions showed no significant findings and negative imaging. The patient subsequently began to develop neurologic symptoms including allodynia and urinary retention.
Ultimately, porphyria urine tests showed elevated porphobilinogens in a pattern that confirmed HCP. The patient was started on a 7 day course of hemin with subsequent resolution of his acute pain crisis, constipation, and urinary retention, but long term persistence of his neuropathy.
Conclusions: This case emphasizes the importance of considering rare disease etiologies such as porphyria on the differential diagnosis for image negative abdominal pain. Porphyria patients can present with nonspecific symptoms and undergo costly work-ups with countless labs and imaging orders, when an inexpensive urine test could quickly confirm the diagnosis. Prompt diagnosis and treatment of acute porphyria episodes is crucial to prevention of prolonged pain episodes, persistent neurologic damage, and death.
An early neurotoxicity assessment tool to optimize management after CAR T-cell therapy
Arvind Suresh1, Heather A Wishart PhD2, Amy M Chan MD3, Bryce Jacobson PsyD2, Grant Moncrief PsyD2, Dorothy McKenna3, Christopher H Lowrey MD3, Christi A Hayes MD3, Kate Caldon RN3, Kenneth R Meehan MD3, and John M Hill Jr MD3
1 Geisel School of Medicine at Dartmouth, 2 Neuropsychology Team, Department of Psychiatry, Dartmouth-Hitchcock Medical Center, 3 Department of Hematology-Oncology, Dartmouth-Hitchcock Medical CenterBackground: Chimeric antigen receptor (CAR) T-cell therapy has transformed treatment of relapsed/ refractory hematologic malignancies, yet carries the risk of significant adverse events, namely cytokine release syndrome and neurotoxicity. In 2018, the American Society for Transplantation and Cellular Therapy (ASTCT) published a consensus grading scale to measure neurotoxicity. Certain neurologic and cognitive-behavioral signs are not included, yet have been observed at our center and also documented in the literature. A more refined assessment tool is therefore needed to capture and grade subtle findings that may facilitate earlier intervention.
Methods: We initiated a prospective study assessing a novel neuropsychological assessment tool to detect early, subtle neurotoxicity following CAR T-cell therapy. Neurologic events were categorized into domains missed by standard criteria (including speech/language, processing speed, fine motor, executive function, attention, working memory, visuo-spatial integration, and personality/mood changes) and mapped to corresponding measures least susceptible to practice effects. During the first 30 days of treatment, the resulting 10-minute battery of tests is administered every four or eight hours, following, and dependent upon, administration of the ASTCT’s standard assessment. The primary objective is to assess the sensitivity/specificity of our tool in detecting clinically relevant changes correlating with impending neurotoxicity.
Results: Upon early review, two tests (Trail Making and Verbal Fluency) appear to show sensitivity for detection of early neurologic changes preceding standard criteria by up to 32 hours. Our tool is easily administered by nurses, and its implementation is feasible. Further conclusions await more definitive data.
Chimeric antigen receptor (CAR) T-cell therapy for hematologic malignancies: Early experience and outcomes from a single, rural-based transplant and cellular therapy center during the COVID-19 pandemic
Arvind Suresh1, Dorothy McKenna MS2, Christopher H Lowrey MD2, Christi A Hayes MD2, Kate Caldon RN2, Lisa Wesinger2, Judi Gentes2, Hali Robinson2, Charlotte Coughenour2, Kenneth R Meehan MD2, and John M Hill Jr MD2
1 Geisel School of Medicine at Dartmouth, 2 Department of Hematology-Oncology, Dartmouth-Hitchcock Medical CenterBackground: Chimeric antigen receptor (CAR) T-cell immunotherapy has transformed treatment of relapsed/refractory hematologic malignancies, with FDA approval of several agents.
Results: Seven patients received CAR T-cell therapy at DHMC (Large B-cell Lymphoma, n=4; Follicular Lymphoma, n=1; Mantle Cell Lymphoma, n=1; Multiple Myeloma, n=1). All tolerated lymphodepletion therapy and CAR T-cell infusion. Hospitalization varied between 14-16 days, and patients were followed locally until Day +30, per FDA mandate.
Most patients experienced mild cytokine release syndrome (CRS; n=5), managed with tocilizumab, an IL-6 receptor antagonist, with or without dexamethasone. Two patients experienced mild Immune Effector Cell Associated Neurotoxicity Syndrome (ICANS), with response to steroids. Two patients required ICU-level care. All CRS and ICANS changes resolved by discharge. Three patients manifested prolonged hypogammaglobulinemia (IgG < 400 mg/dl), responsive to IVIG.
Currently, six patients are beyond 30 days post-therapy. Five patients demonstrated complete remission by PET/CT imaging at Day +30 (n=3), 6 months (n=1), and 1 year (n=1). The Myeloma patient has shown an early marrow response. Current follow-up of these patients reveals no deaths and no evidence of progressive disease, to date.
Conclusions: CAR T-cell therapy is a promising treatment for refractory hematologic malignancies, with demonstrated safety and efficacy in our rural cancer center setting. All patients continue to report an active lifestyle and favorable quality of life in recovery. We are optimistic about the use of this novel modality for patients with hematologic malignancies and, hopefully, solid tumor neoplasms in the future.
Medicaid Expansion Status Affects Locoregional Treatment Modality for Patients with Unresectable Hepatocellular Carcinoma
Benjamin M. Weaver1, Nataniel H. Lester-Coll2, Steven D. Lidofsky2, Geoffrey M. Scriver2
1 Larner College of Medicine, University of Vermont 2 University of Vermont Medical Center, Larner College of MedicineBackground: Locoregional therapies are the primary treatment modality for patients with unresectable hepatocellular carcinoma (HCC) who are not candidates for transplant. Although there are a variety of locoregional modalities available, there are no consensus guidelines dictating which locoregional therapies are most effective for these patients. We sought to describe practice patterns of locoregional therapies in the US and identify factors associated with treatment selection.
Methods: We used data from the National Cancer Database (NCDB) to characterize patterns of usage of various locoregional therapies for patients with HCC from 2004-2017. We categorized locoregional therapies into three groups: ablation (radiofrequency, cryotherapy, or ethanol), transarterial therapies (radioisotopes Radium-223, Strontium-89 and Strontium-90), or stereotactic body radiation therapy. Multivariable logistic regression was used to identify factors associated with treatment modality.
Results: We found the strongest predictor of treatment selection was location in a recent Medicaid expansion state. Patients in states that expanded Medicaid in January 2014 or later were significantly less likely to receive ablative therapies compared to transarterial therapies or SBRT (OR 2.05 and 2.82 respectively, p < .001, 95% CI 1.600 - 1.906) when controlling for year of diagnosis, tumor size, Charlson score, age, race, facility type, insurance status, distance to treatment facility, rurality and income.
Conclusions: Our results suggest that access to Medicaid significantly changed practice patterns with regard to locoregional therapy selection for patients with HCC. Further research is needed to examine whether these changes result in improved patient outcomes.


.png)